Removing Reactive Dyes from Textile Effluent Using Banana Fibre
A.M. Mottakirul Islam
Department of Textile Engineering
Northern University Bangladesh
Email: mishu.aopl@mfgbd.net
Abstract
Use of various dyes in order to color the products is a common practice banana fibre in removing reactive dyes from textile wastewater in the present work in composite knit industry. The presence of these dyes in water even at low concentration is highly visible and undesirable. Initially the adsorbent was tested on standard Remazol Blue RR Reactive dye solutions and then it was also applied to raw textile wastewater. This study was carried out for the utilization of banana fiber as adsorbent for the removal of Reactive dyes from wastewater and to establish it as a standard wastewater treatment process for textile industry. This experiment was performed in the laboratory scale. The materials were obtained and treated for the removal of reactive dyes at different doses. These materials also evaluated for different pH and contact time. This batch adsorption experiment was carried out for finding the effects of adsorbent’s amount, pH and retention time on the removal of dyes from the wastewater. The adsorption was quite fast; approximately 90% of removal efficiency was achieved in 5 mins, and the adsorption was completed in 20 mins under suitable conditions. It was observed that at adsorbent-dye ratio of 333:1, the adsorbent was able to eliminate the reactive dye completely from standard aqueous solution. The equilibrium adsorption behavior was examined by applying adsorption isotherm model. When the adsorbent was applied to raw wastewater containing three reactive dyes Remazol blue RR, Remazol red RR, and Remazol Yellow RR, it was obtained that 1.0 gm of adsorbent was able to convert 30 mL of deep colored wastewater into colorless and transparent water at pH 2. The values of DO, COD, TS, TDS, TSS, salinity and conductivity of wastewater were comparable before and after the interaction with The adsorption comparable to the other available adsorbents.
CHAPTER 1
INTRODUCTION
1.1 Introduction
Textile industry is one of the industries that produce great income to economic growth in several countries around the world. In Malaysia, the textile industry became the country’s 11th largest export income in 2017, which contribute about RM15.3 billion (1.6%) of Malaysia’s total manufactured exports. In 2017, RM428.8 million investment in 12 projects has been approved in the textile and textile industry [1]. However, the wastewater generated contains high organic and inorganic contaminants which exceeds the standard effluent of industrial wastewater by Department of Environment. Untreated textile effluent consists of high pH, temperature, detergents, oil, suspended and dissolved solids, toxic and non-biodegradable matter, color, chemical oxygen demand (COD), and alkalinity [2]. In fact, the textile dyeing consumes large quantity of water and produces large volumes of wastewater from different steps in the dyeing and finishing process [3].
There are more than 100,000 commercially available dyes with 700,000 tons of dye production each year and about 10% of these dyes used in the industry have been lost in industrial effluents [3]. The extensively used of textile dyes in several manufacturing process have been proved to be harmful to the human health as well as to the environment [4]. Several treatment methods had been used such as physical, chemical, and biological methods or suitable combinations of them are available for the treatment of textile wastewater before safely discharged into the environment. Among them, adsorption process has been proven superior compared to other techniques in terms of its simplicity of design, high removal efficiency and ease operation, [4]. Recently, activated carbon is widely used in adsorption process due to porous structure with large surface area and high adsorption capacity. Although it is advantageous as adsorbent media, the high cost of activated carbon caused researchers to find a cheaper alternative.
Application of an agricultural waste as adsorbent media has gained attention by researchers to explore the potential of the adsorbent media in solving textile wastewater problem. Some of agricultural waste adsorbents such as pink guava waste[3], rambutan peel [5], orange peel[6], groundnut shell waste[7], garlic peel[8], pineapple leaf powder[9], coconut husk waste[10] and banana peel[11] used to remove various type of dyes in wastewater treatment. Banana Fibre is an agriculture plant waste which is among the most popular fruit grown in Asia particularly India, Thailand, China, Indonesia, and Malaysia [12]. A few tons per hectare of the banana stem has been estimated annually and this can lead to disposal issues [13]. Several studies on activated banana stem [12;13;14] and banana peel adsorbent [11; 15] have been conducted by the previous researchers. However, the study on agricultural waste using natural banana stem is still limited and need to be explored as an alternative to remove color in textile wastewater. The aim of this study was to investigate the potential use of banana stem as adsorbent media for the removal of color from Methylene Blue (MB) synthetic dyes solution.
1.2 Problem Statement
Waste Water Removing Process is related to Banana fibre. Of any product as there are many seaming operations need to be done to make a garment. Reactive dyes are highly preferred for cotton for their excellent fastness properties and easy application. But problem in reactive dyeing is the low exhaustion properties due to the hydrolysis of reactive dyes with water. This method consumes only a small amount of water and energy. Removal Reactive Dyes are related to Water of Dyeing Production. Banana fiber used in the present work is able to removing reactive dyes efficiently. The removal efficiency of banana fibre was found to be affected by pH of dye solution, contact time, concentration of dye in the solution, and the amount of adsorbent. pH 2 was found the most effective in removing dyes from the aqueous phase, and increased pH results in decreased removal efficiency.
1.3 Objectives of the Thesis
- To determine the Banana fiber adsorbent was obtained from the local sources of Natural fruit tree. Usable dyes are collected.
- To determine the dyeing Process with varying Dyes types.
- To understand the impact of the above parameters on the Treatment process in Production
- To reacts with fibre in presence of alkali and adheres as a part of fibre.
CHAPTER 2
LITERATURE REVIEW
2.1 Literature Review
There are various industries that use dyes as coloring agents, such as textile, paint, pharmaceuticals, beauty products, and food. The release of dyes to water is harmful to the aquatic lives as most dyes are complex in nature and of synthetic origin [1]. It has been estimated that 10-25% of textile dyes are lost during the dyeing process, and 2-20% of such dyes are directly released as aqueous effluents in various environmental constituents [2]. Some of the techniques used to treat dye-containing wastewater are Fenton oxidation, membrane filtration, photo degradation, coagulation-flocculation, biosorption, and adsorption [3–10]. Leading to significant water pollution, a great problem in contemporary time. The World Bank has estimated that almost 20% of global industrial water pollution comes from the treatment and dyeing of textiles [4]. Diseases: In humans, drinking or consuming polluted water in any way has many disastrous effects on our health. It causes typhoid, cholera, hepatitis and various other diseases. Eutrophication: Chemicals in a water body, encourage the growth of algae. In many developing countries particularly in India, China, and Bangladesh, the dye-enriched textile wastewaters are not treated at all, they are just dumped into various water bodies, which are ultimately contributing to environmental degradation, killing aquatic lives, and harming human health [5, 6, 7, 8, 9, 10]. For more than 90% of the earth’s population, water is a limited resource with many regions having less than 10 L/day/person availability. In contrast, the textile processing industry requires 50-150 L/kg of textile material processed [11]. Biological treatment also cannot remove sufficient colored materials. Physio-chemical separation depends on the forces of chemisorption to extract the colloidal organic compounds from the liquid phase [11].
In recent years, the uses of natural adsorbents have gained a remarkable importance due to their low cost, environmental friendliness, local availability, and sustainability [12]. Monika Cherub reported that different adsorbents and commercial activated carbon are preferred sorbents for color removal, however, they are not widely used because of high cost. It is evident from the literature that low cost sorbents have demonstrated outstanding removal capabilities for certain dyes [13]. Many agricultural wastes and natural adsorbents have been tested for the purpose of eliminating the dyes from textile effluents [14, 15]. Wong et al. [16] studied the removal of basic and reactive dyes using sugarcane bagasse. In his study, removal of Basic Blue 3 (BB3) and Reactive Orange 16 (RO16) were studied in single and binary systems. The effect of pH was found noticeable in the sorption of dye, and the optimum pH was determined in the range of 6-8.
Ong et al. [17] studied the removal of basic and reactive dyes using ethylene diamine modified rice hull. Dyes used in that study were Basic Blue-3 (BB3) and Reactive Orange-16 (RO16) in both single and binary systems. Various important parameters such as pH, initial dye concentration, sorption isotherm, agitation rate, particle size, and sorbent dosage were investigated by using batch adsorption studies. The results showed that the sorption of targeted dyes were pH and concentration dependent. The uptake of BB3 was favored at high pH whereas that of RO16 was favored at low pH. An enhancement of 4.5 and 2.4 fold were reported for BB3 and RO16, respectively, in binary dye systems. Pavan et al. [18] studied the removal of Methylene Blue dye from aqueous solution by adsorption using yellow passion fruit peel as adsorbent. It was observed that an alkaline pH was favorable for the adsorption of Methylene Blue, and the maximum adsorption was achieved at 56 hours of contact time. Gupta et al. [19] studied the adsorption of hazardous dye, Erythrosine, over hen feathers. The parameters studied included pH, concentration of dye, temperature, and dosage of adsorbent. The reported optimum pH range for the removal of Erythrosine was 3-8. It also showed that as the temperature was increased, the adsorption of dye was also increased. Weng et al. [20] investigated the removal of Methylene Blue (MB) from aqueous solution by adsorption onto pineapple leaf powder (PLP).
CHAPTER 3
MATERIALS AND METHODS
3.1 Materials
The Banana fiber adsorbent was obtained from the local sources of Natural fruit tree. The usable dyes of Remazol blue RR was collected from dystar (Singapore) Pvt.Ltd. Banani DOHS, Dhaka Cantorment, Dhaka, Bangladesh. Hydrochloric acid and sodium hydroxide use of Chemical was purchased from Merck KGaA, Damstadit, Germany. The Nitrate filter of Cellulose Paper was collected from Biotech, Germany.
3.2 Methods
The bark like part of the banana pseudo-stem was removed from the plant and cleaned with distilled water. Then it was chopped into pieces of about 2 cm × 2 cm, which were boiled in distilled water for two hours. The boiled sample was washed 3 times with distilled and deionized water, and then the sample was dried at 80°C in an oven for 24h. After drying, the sample was ground with a blender (Philips, HR2118). Finally, the ground sample was strained with a sieve shaker (FiltraVibracion SL, Model FLT 200) using a sieve of 212 µ pore size. The appearance of the adsorbent at different stages is shown in Fig 3.1.

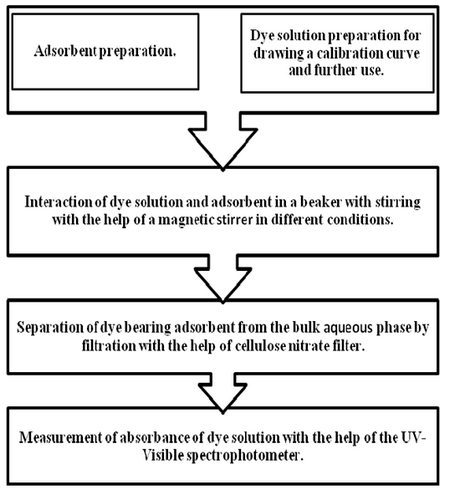
For drawing the calibration curve, a stock solution of Remazol Blue RR dye was prepared by dissolving 0.1 g of dye in distilled water in a 1 L volumetric flask. The stock solution was then diluted by adding distilled and deionized water to make standard solutions of different concentrations. The standard solutions were scanned by a UV- Visible spectrophotometer (Datacolor 850) to find out the λmax of the dye. A calibration curve was drawn by measuring the absorbance of the prepared standard solutions at the obtained λmax. For the interaction of the adsorbent and dye solution, a particular amount of banana fibre was taken in a small beaker and Remazol Blue RR reactive dye solution was added to the beaker. A magnet was inserted in the beaker which was then placed in a magnetic stirrer for a specific time. After that the adsorbent was separated from the aqueous phase by filtration with the help of cellulose nitrate filter. Finally, the dye concentration in the filtrate was measured by the UV Visible spectrophotometer at the obtained λmax. The flow diagram of the experimental setup is shown in Fig. 3.2.
3.3 Preparation of Adsorbent
Collecting
↓
Cutting (2cm x 2cm)
↓
Washing Boiling (2 hours)
↓
Drying (24hrs x 800)
↓
Blending
3.4 Collection of Waste Water
Raw waste water were directly collected from dye bath of Anowara Knit Composite Ltd. Mauna, Sreepur, Gazipur.
3.5 Dyeing Procedure
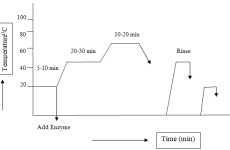
Dyeing procedure Pretreated banana fibers and pretreated cotton fiber samples were used for this dyeing experiment. Four specimens of each 20 grams of banana fibers and cotton fibers were selected from the above pretreated fibers for the dyeing process. Dyeing treatment liquor ratio is 1:50. Oven-dry weight of 20 g each of banana fiber and cotton fiber were dyed with three standard colors (red, blue, and yellow) each with four concentrations (0.25%, 1%, 4%, and 6%). Remazol Yellow RR, Remazol Blue RR, and Remazol Red RR dye types were used and maintained with the concentration of salt at 18 g/L, concentration of soda ash at 3.6 g/L, concentration of wetting agent at 2 g/L. The dyeing temperature was kept at 45°C and gradually raised to 60°C for 90 min.

CHAPTER 4
RESULT AND DISCUSSIONS
4.1 Banana Fibre Adsorbent
Collected banana fibre was boiled with distilled water to remove lignin that can interfere in the measurements. The ground fibre was heterogeneous in size. The ground fiber was sieved to obtain the particles size of ≤ 212 μ for adsorption experiments. Larger particles were avoided due to their less surface area.

4.2 Calibration Curve
For developing the calibration curve for Remazol Blue RR dye, standard dye solutions of 10, 15, 20, 25 and 30 ppm concentrations were prepared. To determine the λmax of the dye, all the solutions were applied for scanning in the range 190-1000 nm with the help of the UV-Visible spectrophotometer. The obtained λmax of the dye was 614 nm. Absorbance of all the standard solutions was measured at 614 nm and the calibration curve (Fig. 4.2) was drawn.

The absorbance of the solvent (i.e. distilled and deionized water) used in these experiments was also measured and it was found that the absorbance at 614 nm was zero. From the curve, it is clear that it is in accordance with the Beer-Lambert law in the used range of concentrations. The value of coefficient of determination (i.e. R2) was 0.999 which establishes the statistical validity of the measurements.Interaction of the adsorbent with the Remazol Blue RR dye and calculation of removal efficiency In a typical experiment, 0.1g of the adsorbent was interacted with 30 mL of dye solution of specified concentration. The dye concentration in the filtrate was calculated from the calibration curve with the help of the obtained absorbance. The removal efficiency of the adsorbent was calculated by using the following formula :
……………………………………..C0 – Ct
Removal efficiency (%) = –––––––––––––––– × 100
………………………………………..C0
Where,
C0 = initial concentration of dye solution (ppm),
Ct = concentration of dye at any time (ppm)
4.3 The Effect of Amount of Adsorbent
The effect of amount of adsorbent was studied in the range of 0.05-0.2 gm of adsorbent when 30 mL of dye solutions of constant concentration (10 ppm) were used for 20 mins. It was observed that as the dosage of adsorbent was increased, the removal efficiency became higher. Increase in the removal efficiency of Remazol Blue RR with the increased amount of adsorbent dose is due to the increased surface area and the availability of additional adsorption sites. It was found that 0.1 g of banana fibre resulted in 100% removal of Remazol Blue RR reactive dye in this particular condition. The adsorbent/dye ratio obtained for quantitative adsorption of dye molecules is approximately.

4.4 Effect of pH
The adsorption of dye molecules on the adsorbent was observed in the pH range 2-12. It exhibited lower removal efficiency in higher pH values. At pH 12, no significant removal took place and it was only about 4%. As the pH value decreases, the removal efficiency is increased and it becomes 100% at pH 2. It is due to the fact that as the pH of the dye solution is decreased, the number of negative charges on surface sites decreased and the number of positively charged surface sites increased, and this favors the adsorption of dye anions due to electrostatic attraction [26]. A similar observation was also seen for the adsorption of Acid Red 183 and Acid Green 25 onto shells of bittim and removal of Acid Blue 62 (AB62) on aqueous solution using calculated colemanite ore waste.

4.5 Effect of Initial Dye Concentration
Different initial dye concentrations ranging from 10-25 ppm were used for adsorption keeping the other parameters (i.e. contact time, pH, amount of adsorbent and amount of dye solution) constant. As the initial concentration of Remazol Blue RR was increased, the absorbance efficiency was decreased. This is because of the fact that the number of dye molecules is increased in the aqueous phase and it is assumed that at 10 ppm concentration, the surface area of the adsorbent becomes saturated, beyond which additional molecules those are present cannot be adsorbed any more resulting to a lower efficiency. Fig. 4.5 shows the effect of initial dye concentration on adsorption. adsorption was completed within 20 minutes. As no dye molecule was observed in the aqueous phase keeping the dye bearing adsorbent up to 60 m, it can be assumed that there was no desorption of dye molecules in that period.


The present adsorbent is found more efficient than orange peel and fly ash whereas it is comparable with modified banana trunk fibres. The present adsorbent takes less time compared to other adsorbents in removing dyes form the bulk aqueous phase.
4.6. Interaction of the Adsorbent with the Raw Wastewater
The sample of raw wastewater was collected from a local textile industry premise and it was preserved in the refrigerator at 4°C. The authority of the industry informed that the collected wastewater contained a blend of three dyes Remazol Blue RR, Remazol Yellow RR, and Remazol Red RR. For characterizing the wastewater, the parameters that were selected are pH, Dissolved Oxygen (DO), Chemical Oxygen Demand (COD), Total Solid (TS), Total Dissolved Solid (TDS), Total Suspended Solid (TSS), salinity, and electrical conductivity. All these parameters were also determined after interacting the wastewater with the. banana fibre. The removal efficiency was also evaluated visually. It was found that 30 mL of deep coloured raw wastewater was converted into colourless and transparent water with the help of 1 gm of banana fibre at pH 2. Experiments were also carried out without adjusting the pH of the raw wastewater and it was found that removal efficiency was much less compared to that with adjusting pH at 2. Fig. 4.7 shows the appearance of the raw wastewater and treated wastewater. The values obtained from characterization before and after the treatment are shown in Table 1.

Table No-1 Physical characteristics of Waste Water before and after Treatment.
| Parameter | Values | |
| Before Treatment | After Treatment | |
| pH | 10.2 | 2.5 |
| DO, ppm | 3.5 | 4.7 |
| COD, ppm | 900 | 850 |
| TS, ppm | 33500 | 33220 |
| TDS, ppm | 31700 | 31620 |
| TSS, ppm | 280 | 13 |
| Salinity , ppm | 22000 | 21000 |
| Conductivity, ms/cm | 34.6 | 36 |
The alkaline wastewater was converted into acidic water; this is basically due to adjustment of pH at 2 which is the obtained optimum pH for the current adsorption system. The adsorption results in approximately 20% increase in DO level whereas it does not result in significant improvement in COD. The amount of TS, TDS and TSS were found to decrease due to adsorption. The salinity of water before and after the adsorption was comparable. This is due to the fact that the number of ions is increased due to the adjustment of pH to 2. Similarly, there was no remarkable change in electrical conductivity.
CHAPTER 5
CONCLUSION
5.1 Conclusion
Banana fibre used in the present work is able to remove Remazol Blue RR, Remazol Yellow RR, and Remazol Red RR reactive dyes efficiently. The removal efficiency of banana fibre was found to be affected by pH of dye solution, contact time, concentration of dye in the solution, and the amount of adsorbent. pH 2 was found the most effective in removing dyes from the aqueous phase, and increased pH results in decreased removal efficiency. The adsorption is very quick, and the quantitative adsorption occurs within 20 mins.
Therefore, it can be concluded that the process is relatively less time consuming. The Adsorbent/dye ratio is important for the present system and quantitative adsorption is achieved at the adsorbent/dye ratio of 333:1. The adsorbent is also found able to remove reactive dyes from raw wastewater efficiently. The treatment with the present adsorbent leads to a significant decrease in pH which needs to be adjusted with the help of an alkali before discharging treated wastewater into natural surface water systems. The present study suggests that the banana fibre can be used as a sustainable adsorbent to remove various reactive dyes from textile effluents efficiently.
5.2 References
[1] Ogata, D. Imai, and N. Kawasaki, “Cationic dye removal from aqueous solution by waste biomass produced from calcination treatment of rice bran,” Journal of Environmental Chemical Engineering, vol. 3, no. 3, pp. 1476–1485, 2015.
[2] http://cdn.intechopen.com/pdfs-wm/29369.pdf (Accessed Jan. 2013).
[3] Wang, Y. Guan, L. Wang et al., “Fabrication of a novel bifunctional material of BiOI/Ag3VO4 with high adsorption– photocatalysis for efficient treatment of dye wastewater,” Applied Catalysis B: Environmental, vol. 168-169, pp. 448–457, 2015.
[4] http://www.sustainablecommunication.org/eco360/ what-is-eco360s-causes/water- pollution (Accessed Nov. 2014).
[5] http://www.theguardian.com/sustainable-business/dyeing-textilesector-water-risks- adidas (Accessed Jan. 2014).
[6] M. Islam, K. Mahmud, O. Faruk, and M. S. Billah. (2011). Textile dyeing industries in Bangladesh for sustainable development. International Journal of Environmental Science and Development. 2(6).
[7] https://www.dosomething.org/facts/11-facts-about-pollution (Accessed Jan. 2014).
[8] http://www.fibre2fashion.com/industryarticle/43/4229/biodegradation-of-textile- wastewater2.asp (Accessed Jan. 2014).
[9] http://www.nmglyt.com/effects-of-water- pollution-an-uprisingconcern
[10] T.Sarat Chandra, S.N.Mudliar, S.Vidyashankar et al., “Defatted algal biomass as a non-conventional low-cost adsorbent: surface characterization and methylene blue adsorption characteristics,” Bioresource Technology, vol. 184, pp. 395–404, 2015.
[11] Support to the Bangladesh quality support program, textiles and RMG component, the treatment of textile effluent – the current status with particular reference to Bangladesh, Park, J., UNIDO report, 2011.
[12] B. A. Rahman, M. Akter, and M. Z. Abedin. (2013). Dyes Removal From Textile Wastewater Using Orange Peels. International Journal of Scientific & Technology Research. 2(9), pp. 47-50.
[13,14] S. Y. Wong, Y. P. Tan, A. H. Abdullah, and S. T. Ong. (2009). The removal of basic and reactive dyes using quartenised sugar cane bagasse. Journal of Physical Science. 20(1), pp. 59-74.
[15] R. Mafra, L. Igarashi-Mafra, D. R. Zuim, E. C. Vasques, and M. A. Ferreira. (2013). Adsorption of Remazol Brilliant Blue on an orange peel adsorbent. Brazilian Journal of Chemical Engineering. 30(03), pp. 657 – 665.
[16] Y. Wong, Y. P. Tan, A. H. Abdullah, and S. T. Ong (2009). The removal of basic and reactive dyes using quartenised sugar cane bagasse. Journal of Physical Science. 20(1), pp. 59-74.
[17] T. Ong, C. K. Lee, and Z. Zainal. (2008). Ethylene diamine modified rice hull as a sorbent for the removal of Basic Blue 3 and Reactive Orange International Journal of Environment and Pollution. 34, pp. 1-4.
[18] A. Pavan, A. C. Mazzocato, and Y. Gushilem. (2008). Removal of Methylene Blue from aqueous solutions by adsorption using yellow passion fruit peel as adsorbent. BioSource Technology. 99, pp. 3162-3165.
[19] K. Gupta, A. Mittal, L. Kurup, and J. Mittal. (2006). Journal of Colloid and Interface Science. 304, pp. 52-57.
[20] H. Weng, Y. T. Lin, and T. W. Tzeng. (2009). Removal of Methylene Blue from aqueous solution by adsorption onto pineapple leaf powder. Journal of Hazardous Materials. 170, pp. 417-424.
APPENDIX








You may also like:
- Methods for Wastewater Treatment in Textile Dyeing Industry
- Water Management in Textile Industry – An Overview
- Reduction of ETP Load through Wastewater Segregation
- Implementation of Effluent Treatment Plants for Wastewater and Effect of Untreated on Environment
- Banana Fiber: Properties, Manufacturing Process and Applications
- Reactive Dyes: Classification, Dyeing Mechanism, Application & Stripping
Founder & Editor of Textile Learner. He is a Textile Consultant, Blogger & Entrepreneur. Mr. Kiron is working as a textile consultant in several local and international companies. He is also a contributor of Wikipedia.