Practices Towards Green Apparel Product: Investigation on Environmental Issues at Different Stages of Knitted T-shirt Development
Md. Zahid Hassan, Md. Suruj-Zaman &
Zinia Anjuman Ara
Department of Textile Engineering
Dhaka University of Engineering & Technology (DUET)
Gazipur -1700, Bangladesh
ABSTRACT
In this study, the ecological parameters of the effluents obtained from scouring and dyeing with reactive dyes of Remazol brand for conventional cotton single jersey knitted fabric as well as bio-scouring and dyeing with low impact reactive dyes of Avitera brand for organic cotton single jersey fabric have been investigated. The ecological parameters include chemical oxygen demand (COD), biological oxygen demand (BOD), total dissolved solids (TDS), dissolved oxygen (DO), EC or conductivity, alkalinity and pH. Also, some selected physical properties have been investigated for both dyed fabrics such as bursting strength, fabric drapability, color fastness to wash, color fastness to rubbing and color fastness to perspiration. Finally, T-shirt has been made with both fabrics to show and enhance eco-friendly product development. The results of ecological test show greater ecological advances for using organic cotton, bio scouring and low impact reactive dyes than the conventional method and raw materials. The physical properties also show better results of using eco-friendly fabric and processes. Finally, the use of small printing area on the organic cotton T-shirt with low impact reactive dye has also enhanced the environmental sustainability of the apparel product. Moreover, a hang tag has been developed which provides care instructions as well as aware the retailers and customers to accept green apparel product. Finally, by using Higg index environmental performance of both the t-shirt has been investigated. The result of Higg index shows that the T-shirt made from organic cotton has lower environmental impacts than the T-shirt made from conventional cotton.
Key words: Organic cotton, Conventional cotton, Environment friendly, Ecological Parameters, Green apparel product, Higg Index.
1. INTRODUCTION
1.1 Introduction
Developing economies together with the increasing global population are having a direct impact upon production and consumption of both fossil and natural resources. This has the potential to lead to further significant environmental degradation if appropriate measures are not put in place to control, manage or mitigate the impact of such developments. There is a need wherever possible to dissociate growth from increased consumption of fossils fuels, water, energy and reduce the use and resulting impact upon natural resources. Output to the environment from production and waste must also be reduced in volume and impact (R.S. Blackburn, 2009).
Sustainability is the major concern in the age of modern world. And for textile and apparel sector, this has been a burning issue for many related concerned bodies. Sustainable development indicates the development that meets present needs without compromising future generations’ welfare (World Commission on Environment and Development, 1987). From an industry perspective, sustainable development means that “the economic bottom line is congruent with improving social conditions and environmentally responsible manufacturing because (i) a motivated, healthy workforce living in a thriving community is more productive and (ii) using less resources, generating less waste, and improving quality reduces costs while increasing demand” (Mihelcic et al., 2003).
Rapid growth in the industrial sector is playing a vital role in the economy of Bangladesh. Mainly the growth has been concentrated in garments which are export oriented industries. To support garments a large number of other textile industries have been established & more are growing to be set up shortly. The textile industry actually represents a range of industries with operations and processes as diverse as its products (Jayanth et al, 2011; Sajjala et al, 2008). Greige fabrics, after its manufacturing, are subjected to several wet processes such as pretreatment process involving demineralization, scouring, bleaching and mercerization. The pretreated fabric is then dyed using textile dyes and finished by softener padding. The pretreatment and dyeing process results in large volume of effluent that has harmful effect on environment (Correia et al 1994; Desai &Kore, 2011; Sofia et al, 2000; Sajjad & Amna, 2011; Joshi & Santani, 2012; Lav & Jyoti, 2011; Sivakumar. et. al, 2011, Ramesh et al, 2007; Sengupta, 2007; Tufekci et al, 2007; Bisschops & Spanjers, 2003; Imtiazuddin et al, 2012). The high volume of effluent is primarily due to large volume of water and chemicals used during wet processing. The chemicals used in textile sector are diverse in chemical composition ranging from inorganic to organic. Generally, textile effluent is colored with high pH, temperature, biological oxygen demand (BOD), chemical oxygen demand (COD), total dissolve solid (TDS) and total suspended solid (TSS). Color is imparted to textile effluents because of the various dyes and pigments used to color the fabric. The presence of dyes in the waste waters will cause severe damage to the aquatic biology. This is because dyes have a synthetic origin and a complex molecular structure which makes them more stable and difficult to be biodegraded (Noor & Rohasliney, 2011).
It is estimated that (12-65) L of water is required for processing one meter of cloth. So, it is easily understood that what a large volume of waste water generates daily from different textile mills. In 1943 it was estimated that from 502 textile industries 40,000 m3 /day waste water was discharged in Bangladesh which created 26,000 kg/day BOD load. According to another report individual small-scale dyers discharge 8m3/day waste water and the corresponding value for large scale dyers is 70-400 m3/day.
1.2 Purpose and Significance
To minimize the above mentioned causes of environmental pollution of our country as well as the whole world this thesis is a little approach. This thesis contains the investigation of the environmental pollution of conventional cotton knit fabric processing from pretreatment to T-shirt production and organic cotton knit fabric processing using enzymatic scouring and low impact reactive dyes to apparel production. Some selected physical properties of both type of finished fabric have been studied to do a comparative study. Finally it is intended to encourage the consumer to use green product by providing conscious messages through a hang tag which is the approach to move the thinking of the customer towards sustainability.
1.3 Aims and Objectives of the Research
- To select the materials and manufacturing process issues in the development of eco-friendly or green apparel product i.e. T-shirt.
- To investigate the environmental impact by effluents of conventional and organic cotton single jersey knit fabric on different stages of wet processing.
- To investigate some selected physical properties of both organic and conventional cotton single jersey knit fabric.
- Printing of T-Shirt focusing environmental issues.
- Developing a hang tag with maximum information transparency for manufacturers, stakeholders and consumers to aware them about green apparel products consumption.
- Finally developing a T-shirt by considering possible environmental issues.
- Evaluation of environmental performance of newly developed T-shirt by Higg Index (A standard assessment tool by sustainable apparel coalition, USA)
1.4 Research Questions:
- Is it possible to get same or better appearance and fastness properties using fabric of organic cotton as compared with fabric of conventional cotton?
- Is it possible to reduce environmental impacts using eco friendly pretreatment chemicals and low impact reactive dye?
- Is the enzymatic scouring process and dyeing process with low impact reactive dye production friendly as compared with caustic scouring process and dyeing with typical reactive dye respectively?
- What is the proportion of environmental impact to make an eco-friendly t-shirt in place of using conventional cotton and process to make a conventional t-shirt?
2. LITERATURE REVIEW
2.1 Literature Review
The textile industry is shared between natural fibres such as wool, silk, linen, cotton and hemp, and man-made ones, the most common of which are synthetic fibres (polyamide, acrylic) made from petrochemicals. Most of the clothes in our wardrobes contain polyester, elastane or Lycra. These cheap and easy-care fibres are becoming the textile industry’s miracle solution. However, their manufacture creates pollution and they are hard to recycle (for example with nylon taking 30 to 40 years to decompose). The textile and clothing industry is a diverse one, as much in the raw materials it uses as the techniques it employs. (Environmental Impact of Textiles: Production, Processes and Protection by Keith Slate) When the actual ways in which textile goods are made are put under close scrutiny, plenty of instances of environmental concern can be found. These will be looked at in sequence, following the various stages in turn, from growing or manufacturing the starting materials, the fibres, to the point at which an end product is shipped to the final consumer in readiness for use.
An anonymous author records annual world production of textile fibres at about 60 million tonnes, of which over 50% is synthetic. This is in agreement with another writer, who quotes a value for world production of textile fibres of almost 58 million tonnes, with chemical fibres amounting to 31 million tones. Any growth likely to take place will probably be in the latter type of textiles; Schenek surveys comprehensively the state of natural fibre production and notes that, although the cotton harvest grew by almost 50% in the last decades of the 20th century, the production of other natural fibres is likely to remain more or less steady. His estimate of total natural fibre production is in slight disagreement with the figures quoted above, since he records it at only about 26% of world total.
Sustainable Apparel Product
Sustainability is defined as the design of human and industrial systems to ensure that humankind’s use of natural resources and cycles do not lead to diminished quality of life due either to losses in future economic opportunities or to adverse impacts on social conditions, human health and the environment” (Mihelcic et al., 2003, p.5315). To achieve sustainable development, designers need to be aware of environmental impacts and incorporate environmental awareness into the design (Holdway, Walker, & Hilton)
Sustainable clothing refers to fabrics derived from eco-friendly resources, such as sustainably grown fiber crops or recycled materials. It also refers to how these fabrics are made. Historically, being environmentally conscious towards clothing meant (1) buying clothes from thrift stores or any shops that sell second-hand clothing, or (2) donating used clothes to shops previously mentioned, for reuse or resale. In modern times, with a prominent trend towards sustainability and being ‘green’, sustainable clothing has expanded towards (1) reducing the amount of clothing discarded to landfills, and (2) decreasing the environmental impact of agro-chemicals in producing conventional fiber crops (e.g. cotton). Under the accordance of sustainability, recycled clothing upholds the principle of the “Three R’s of the Environment”: Reduce, Reuse, and Recycle, as well as the “Three Legs of Sustainability”: Economics, Ecology, and Social Equity (wikipedia, 2014).
Theodore Roszak in his book ‘The Voice of the Earth’ (Roszak, 1993), thinks that if psychosis is an attempt to live a lie, then our psychosis is believing we have no ethical obligation to our planetary home.
The following five headings describe the issues that make the life-cycles of textiles and clothing unsustainable. If we include issues of fair-trade and ethical treatment of workers the list is even longer.
- Water. Misappropriation and inappropriate use. Excessive use. Contamination.
- Chemicals. Profligate use of pesticides and herbicides in agriculture and of toxic chemicals in production.
- Asset stripping of non-renewable resources, including energy sources. Undervaluation and non-regulation.
- Waste. Too much is destroyed. Systems need to be developed to recycle all non-renewables or compost renewables.
- Transport. Unnecessary demand caused by capitalist exploitation of cheap labour makes this also unethical as well as increasing the profligate use of non-renewable resources such as oil, and the attendant pollution (A. SHERBURNE, 2011).
Patagonia described what they considered their “ideal garment” (Brown & Wilmanns, 1997), adhering to their mission of “‘maximum attention to product quality’ while ‘striving to do no harm’ to the environment” (Chouinard & Brown, 1997, p. 118). Patagonia’s LCA outline (Table 2.1) is used to organize the major environmental concerns of the clothing life cycle according to these six life stages.
Table 2.1: Patagonia’s Definition of ‘Ideal Garment’ (Brown & Wilmanns, 1997)
| Product design criteria |
|
| Materials selection |
|
| Production processes |
|
| Distribution |
|
| Product maintenance |
|
| End of life |
|
2.2 Environmental Effects of Textile Fibres
2.2.1 Cotton
Cotton is the most pesticide intensive crop in the world: these pesticides injure and kill many people every year. It also takes up a large proportion of agricultural land, much of which is needed by local people to grow their own food. Herbicides, and also the chemical defoliants which are sometimes used to aid mechanical cotton harvesting, add to the toll on both the environment and human health. These chemicals typically remain in the fabric after finishing, and are released during the lifetime of the garments. The development of genetically modified cotton adds environmental problems at another level. Growing cotton uses 22.5% of all the insecticides used globally. Growing enough cotton for one T-shirt requires 257 gallons of water. On top of that, bleaching and then dyeing the resulting fabric creates toxins that flow into our ecosystem.
First of all, the cotton must be grown; this entails vast amounts of chemical fertilizers and pesticides that pollute and deplete the soil. Despite mechanized harvesting, the cotton industry is still largely dependent on cheap labour. The raw cotton is then dyed, meaning chemicals and heavy metals with harmful effects on the environment. Finally, bands of cotton are assembled in factories to be sewn into a T-shirt. From waste water emissions to air pollution and energy consumption, the textile industry weighs heavily on the environment.
2.2.2 Wool
Both agricultural and craft workers in the UK suffer from exposure to organophosphate sheep dip problem. Getting from fibre to cloth – bleaching, dyeing, and finishing – uses yet more energy and water, and causes yet more pollution.
2.2.3 Nylon and polyester
They are made from petrochemicals, these synthetics are also non-biodegradable, and so they are inherently unsustainable on two counts. Nylon manufacture creates nitrous oxide, a greenhouse gas 310 times more potent than carbon dioxide. Making polyester uses large amounts of water for cooling, along with lubricants which can become a source of contamination. Both processes are also very energy-hungry.
2.2.4 Rayon (viscose)
Another artificial fibre is made from wood pulp, which on the face of it seems more sustainable. However, old growth forest is often cleared and/or subsistence farmers are displaced to make way for pulpwood plantations. Often the tree planted is eucalyptus, which draws up phenomenal amounts of water, causing problems in sensitive regions. To make rayon, the wood pulp is treated with hazardous chemicals such as caustic soda and sulphuric acid. The use of rayon for clothing is contributing to the rapid depletion of the world’s forests. Petroleum-based products are detrimental to the environment on many levels.
2.2.5 Asbestos fibres
Modified natural fibre asbestos, has a special place in the environmental debate. Long regarded as a wonder material for its good thermal and electrical insulation abilities, it has been recognized as a dangerous substance because of its tendency to cause lung cancer.
2.3 Selection of Organic Cotton
Organic cotton represents a return to safe and sustainable practices. It is grown with natural fertilizers and is free from toxic chemicals. Organic farmers rely on crop rotation to replenish and maintain soil fertility. Mechanical cultivation and botanical or biological means are used to control pests and weeds. A field must be pesticide-free for at least three years to be certified organic, and the cotton must be processed according to international organic standards. Third-party organizations certify that organic cotton farms use only these approved methods and do not spray toxic chemicals on their crops (HAE Now Organic Tee, 2013).
Some of the benefits of organic cotton are:
- Manual farming and organic practices have a lower carbon footprint as the entire process consumes less fuel and energy and emits fewer greenhouse gases.
- Not from genetically modified cotton seed.
- Grown with natural rather than synthetic pesticides and fertilizers, no chemical defoliants used.
- Eco-friendly processing that does not compromise workers’ health and helps reduce water and electric use and toxic runoff, e.g. non-chlorine bleach, silicon-free softeners and low impact, azo-free dyes.
- Strict testing ensures the absence of contaminants like nickel, lead, formaldehyde, amines, pesticides and heavy metals.
- People with allergies and chemical sensitivity especially get benefits from organic cotton clothing, as conventional cotton may retain harmful toxic residues. Even if one doesn’t have sensitive skin, organic cotton will just feel better against one’s skin.
Organic cotton Vs. Conventional Cotton
- There are a number of important differences between conventional and organic cotton. Starting from the tilling of the soil to the selection of seeds, labor paid and water used, organic cotton farming requires significant investment of time and resources to radically overhaul conventional cotton growing methods.
- Cotton is a crop that attracts a wide range of insects (e.g. cutworm, cotton bollworm, tobacco bollworm, army worm, loopers, aphids, whitefly, spider mite among others). This is one of the reasons why it is one of the largest pesticide dependent crops in the world.
The following table outlines some of the major differences at each stage of the growing process.
Table 2.2: Organic cotton Vs. Conventional Cotton
| Topic | Organic | Conventional |
| Seeds | GMO-free; untreated | Treated with fungicides, insecticides. Possible Genetic Modification |
| Soil | Crop rotation; cover cropping, better retention of moisture; higher concentrations of organic matter, animal manure additions | Synthetic fertilizers, loss of soil due to mono crop culture, intensive irrigation |
| Weed & insect control | Beneficial insects and ‘trap crops’ used; flame weeding and cultivation; Border management of vegetation (to increase population of native beneficial); use of certain bio-pesticides such as bacteria, virus and fungal insect pathogens | Aerial spraying of insecticides used & pesticides; FYI 9 of the most commonly pesticides are known cancer-causing agents |
| Harvesting | Natural defoliation from freezing temperatures or through the use of water management | Defoliation induced with toxic chemicals |
| Whitening | Safe peroxide is used | Chlorine bleaching is used; toxic by product produced and released into environment in process |
| Finishing | Soft scour in warm water with soda ash for pH 7.5-7.8 | Hot water, synthetic surfactants, additional chemicals (sometimes formaldehyde) |
| Dyeing | Low impact or natural dyes used with low metal or sulfur content | High temperature with heavy metals and sulfur content (can leach into skin as well as ecosystem) |
| Printing | Low impact, water-based inks and/or pigments with no heavy metals | Pigments may be petroleum based and contain heavy metals. Run off easily spills into water ways and ecosystem |
| Fair trade | Organic certification includes requirements for fair wages and treatment of workers in the supply chain. Criteria are in place to ensure safe, healthy and non-abusive, non-discriminatory compensation for workers. | No fair trade or living wage requirements in place. Widespread evidence of child labor, forced labor, slave labor/wages and work condition in countries where cotton growing exists. |
2.4 Ecological Parameters
2.4.1 Chemical Oxygen Demand (COD)
The COD is used as a measure of oxygen equivalent of organic matter content of sample that is susceptible to oxidation by strong chemical oxidant for sample from a specific source. Most applications of COD determine the amount of organic pollutants found in surface water (e.g. lakes and rivers) or wastewater, making COD a useful measure of water quality. It is expressed in milligrams per liter (mg/L) also referred to as ppm (parts per million), which indicates the mass of oxygen consumed per litre of solution.
2.4.2 Biochemical Oxygen Demand (BOD)
Biochemical oxygen demand or BOD is the amount of dissolved oxygen needed by aerobic biological organisms in a body of water to break down organic material present in a given water sample at certain temperature over a specific time period. The term also refers to a chemical procedure for determining this amount. This is not a precise quantitative test, although it is widely used as an indication of the organic quality of water. The BOD value is most commonly expressed in milligrams of oxygen consumed per litre of sample during 5 days of incubation at 20 °C and is often used as a robust surrogate of the degree of organic pollution of water (Wikipedia, 2014).
BOD can be used as a gauge of the effectiveness of waste water treatment plants. It is listed as a conventional pollutant in the U.S. Clean Water Act.
BOD is similar in function to chemical oxygen demand (COD), in that both measure the amount of organic compounds in water. However, COD is less specific, since it measures everything that can be chemically oxidized, rather than just levels of biologically active organic matter.
2.4.3 Dissolved Oxygen (DO)
Oxygen saturation in the environment generally refers to the amount of oxygen dissolved in the soil or bodies of water. Environmental oxygenation can be important to the sustainability of a particular ecosystem. A well-mixed body of water will be fully saturated, with approximately 10mg/L at 15 °C. The optimal levels in an estuary for Dissolved Oxygen (DO) are higher than 6 ppm. Insufficient oxygen (environmental hypoxia), often caused by the decomposition of organic matter and/or nutrient pollution, may occur in bodies of water such as ponds and rivers, tending to suppress the presence of aerobic organisms such as fish. Deoxygenation increases the relative population of anaerobic organisms such as plants and some bacteria, resulting in fish kills and other adverse events. The net effect is to alter the balance of nature by increasing the concentration of anaerobic over aerobic species (Wikipedia, 2014).
2.4.4 Electrical conductivity (EC)
The conductivity (or specific conductance) of an electrolyte solution is a measure of its ability to conduct electricity. The SI unit of conductivity is siemens per meter (S/m).
Conductivity measurements are used routinely in many industrial and environmental applications as a fast, inexpensive and reliable way of measuring the ionic content in a solution. For example, the measurement of product conductivity is a typical way to monitor and continuously trend the performance of water purification systems (Wikipedia, 2014).
In many cases, conductivity is linked directly to the total dissolved solids (TDS). High quality deionized water has a conductivity of about 5.5 μS/m, typical drinking water in the range of 5-50 mS/m, while sea water about 5 S/m (i.e. sea water’s conductivity is one million times higher than that of deionized water).
2.4.5 Alkalinity
Alkalinity is the name given to the quantitative capacity of an aqueous solution to neutralize an acid. Measuring alkalinity is important in determining a stream’s ability to neutralize acidic pollution from rainfall or wastewater. It is one of the best measures of the sensitivity of the stream to acid inputs. There can be long-term changes in the alkalinity of rivers and streams in response to human disturbances (Wikipedia, 2014).
2.4.6 Total Dissolved Solid (TDS)
Total dissolved solid (TDS) is a measure of the combined content of all inorganic and organic substances contained in a liquid in molecular, ionized or micro-granular suspended form. Generally, the operational definition is that the solids must be small enough to survive filtration through a filter with two-micrometer (nominal size, or smaller) pores. Total dissolved solids are normally discussed only for freshwater systems, as salinity includes some of the ions constituting the definition of TDS. The principal application of TDS is in the study of water quality for streams, rivers and lakes, although TDS is not generally considered a primary pollutant (e.g. it is not deemed to be associated with health effects) it is used as an indication of aesthetic characteristics of drinking water and as an aggregate indicator of the presence of a broad array of chemical contaminants where TDS is expressed in mg/L (Wikipedia, 2014).
Primary sources for TDS in receiving water are agricultural and residential run off, leaching of soil contamination and point source water pollution discharge from industrial or sewage treatment plants. The most common chemical constituents are calcium, phosphates, nitrates, sodium, potassium and chloride.
Total dissolved solids (TDS) are differentiated from total suspended solids (TSS), in that the latter cannot pass through a sieve of two micrometers and yet are indefinitely suspended in solution. The term “settleable solids” refers to material of any size that will not remain suspended or dissolved in a holding tank not subject to motion, and excludes both TDS and TSS. Settleable solids may include larger particulate matter or insoluble molecules.
2.4.7 pH
In chemistry, pH is a measure of the acidity or basicity of an aqueous solution. Solutions with a pH less than 7 are said to be acidic and solutions with a pH greater than 7 are basic or alkaline. Pure water has a pH very close to 7 (Wikipedia, 2014).
Mathematically, pH is the negative logarithm of the activity of the (solvated) hydronium ion, more often expressed as the measure of the hydronium ion concentration.
2.5 Scouring
Natural fibers contain oils, fats, waxes, minerals, leafy matter and motes as impurities that interfere with dyeing and finishing. Synthetic fibers contain producer spin finishes, coning oils and/or knitting oils. Mill grease used to lubricate processing equipment mill dirt, temporary fabric markings and the like may contaminate fabrics as they are being produced. The process of removing these impurities is called Scouring. Even though these impurities are not soluble in water, they can be removed by Extraction, dissolving the impurities in organic solvents, Emulsification, forming stable suspensions of the impurities in water and Saponification, Converting the contaminates into water soluble components (Tomasino, 1992).
2.6 Bio Scouring
Bio-scouring with pectinases (enzyme) have shown promise in replacing the traditional alkaline scouring treatment. Some auxiliaries’ suppliers have introduced an enzymatic process to remove hydrophobic and other non-cellulosic components from cotton. The new bio-scouring process operates at mild pH conditions over a broad temperature range and can be applied using equipment such as jet machines. It is claimed that, due to a better bleach ability of enzyme-scoured textiles, bleaching can be carried out with reduced amounts of bleaching chemicals and auxiliaries. Bio-scouring enzymes actually make the substrate more hydrophilic (which could explain better bleachability).
2.7 Enzyme
Enzymes are large biological molecules responsible for the thousands of metabolic processes that sustain life. They are highly selective catalysts, greatly accelerating both the rate and specificity of metabolic reactions, from the digestion of food to the synthesis of DNA. Most enzymes are proteins, although some catalytic RNA molecules have been identified. Enzymes adopt a specific three-dimensional structure and may employ organic (e.g. biotin) and inorganic (e.g. magnesium ion) co-factors to assist in catalysis.
Conventional pretreatment process consumes large amount of time, energy and water. The conventional highly alkaline preparation of cotton is an example of a wet chemical process having a negative environmental impact. The greatest problem occurring during conventional pretreatment process with caustic & peroxide is in the radical reactions of the bleaching compounds with the fibre thus reduce the tensile strength of the fabric.
As a remedy, DuPont industrial Bioscience introduces the use of enzymes (Prima Green EcoScour) for bio-scouring. Because of same application condition the Neutral Bio-Polish enzymes (Primafast Gold RSL) can be applied with this process.
2.7.1 Prima Green® EcoScour
Prima Green® EcoScour is a concentrated liquid formulation containing a pH neutral pectate lyase enzyme. This enzyme belongs to the family of lyases, specifically those carbon-oxygen lyases acting on polysaccharides.
Bioscouring is the use of pectate lyase combined with detergents/wetting agents to remove non-cellulosic impurities from cotton fibres. Bio scouring enhances cotton wettability and absorbency.
Pectate lyase hydrolyzes pectin into soluble compounds facilitating the removal of other impurities (e.g. wax and protein) from the cotton during the bioscouring process.
Prima Green® EcoScour enzyme has the following benefits:
- Medium temperature conditions resulting in energy savings/cost savings;
- Mild neutral pH processing conditions resulting in improved fabric weight quality (softer, bulkier hand), lower fabric weight loss, enhanced absorbency and improved fabric dyeability;
- Excellent fit with today’s global green cotton, eco-friendly, worker-friendly and more sustainable processing requirements.
2.7.2 Primafast® GOLD RSL
Primafast® GOLD RSL is a ready-to-use enzyme formulation for textile bio-polishing in mills and laundries. This unique product provides surface defibrillation yielding superior fabric and garment cleanup under neutral pH conditions in all types of processing equipment. Primafast® GOLD can perform at lower than conventional process temperatures and dyeing can follow directly after Primafast® GOLD treatment in the same bath, thus saving time, energy and water. This is bio-degradable.
Primafast® GOLD RSL provides these processing and fabric quality benefits:
- No pH adjustment.
- Combined finishing and dyeing.
- Preserved fabric strength and weight.
- Increased dye uptake.
- Reduced water heating time.
- Combined bio-finishing with Prima Green® Technology.
- Consistent garment quality.
- Increased color brilliance.
2.8 Dyestuffs
Vinyl Sulfone dyes, also known as Remazol® dyes after the trademarked name under which they were first introduced, are a type of fiber reactive dye that is often used in cotton dyeing. Vinyl sulfone dyes are a type of fiber reactive dye that is less reactive than, for example, Procion MX dyes, and thus they both last longer in solution in water, and require more heat for the reaction with the fiber. This means that they can actually be purchased already dissolved in water, unlike Procion MX or Cibacron F dyes, eliminating the one slightly hazardous step of working with powdered dyes.
The AVITERA dye system is economically comparable to conventional dye systems when considering time, energy required (dyeing temperature), dye costs, and water consumption, providing an economically sustainable alternative while also considering an environmental improvement compared to present technology. Making a full color palette of dyes available, Huntsman International developed this new chemistry with the focus on environmental sustainability, and especially to address a more sustainable approach to water use.
AVITERA dyes use tri-functional reactivity that provides a high reaction and fixation rate with cellulosic fiber, leaving very little unfixed dye to be removed, dramatically reducing water waste. Dye that is not reacted is easily removed at low temperatures because the dyes have low intrinsic affinity for the fiber. When using these dyes, up to 50% less water is required, up to 70% less energy is required, and up to 50% less time is used in the dye process.
2.9 Printing and Hangtag
Printing is done in a small area to maintain sustainability with low impact reactive dye.
A hangtag provides sufficient care, reuse and recycle information. It also encourages and aware the retailers and consumers to think about the world’s environment, to think green and to move towards sustainability.
2.10 Higg Index (Rapid Design Module, RDM – Beta)
The RDM-Beta is a prototype designed to test the concept of a simple tool that aims at educating and providing quick directional guidance to apparel and footwear designers during the product creation process about the potential environmental impacts of their design solutions. It is a streamlined version of the Product Module in the Higg Index 1.0 to help engage designers in the Higg Index’s produce life cycle thinking and how we directionally assess materials sustainability through the MSI (Sustainable Apparel Coalition, 2012).
Here is a screenshot to give a sense of what the RDM-Beta looks like:

3. RESEARCH DESIGN
3.1 Diagram of methodology

3.2 Raw Materials
The following raw materials were used during the progress of this thesis and research work.
3.2.1 Fabric
Grey single jersey knitted fabric of organic cotton
Fabric specification:
- Fiber type: 100% organic cotton
- Brand: CISLAND
- Fabric type: Single jersey
- Count of yarn: 38S comb
- Grey GSM: 120
- Finished GSM: 135
- Stitch length: 2.65mm
- Machine Día: 30”
- Machine gauge: 24 G
- CPC: 29
- WPC: 10
- Lot No.: 90020
- Factory: Paul Maul Group
Grey single jersey knitted fabric of conventional cotton
Fabric specification:
- Fabric type: Single jersey
- Count of yarn: 30s
- Grey GSM: 130
- Finished GSM: 150
- Stitch length: 3.15 mm
- Machine dia: 32 inches
- CPC: 18
- WPC: 15
- Factory: Knitting lab of DUET
3.2.2 Chemicals
Table 3.1: List of pretreatment chemicals
| Process | Chemicals | Brand/Origin |
| Bio-scouring of Single jersey knitted fabric of organic cotton | Prima Green Eco Scour | Prima Green, US |
| Prima Fast Gold RSL | Prima Green, US | |
| Detergent | Resotex-DFM, India | |
| Wetting Agent | Resotex-JSSA, India | |
| Scouring of Single jersey knitted fabric of conventional cotton | NaOH | India |
| Detergent | Resotex-DFM, India | |
| Wetting agent | Resotex-JSSA, India | |
| Sequestering agent | Resotex-600S, India | |
| Antifoaming agent | India | |
| CH3COOH | India |
Table 3.2: List of dyes,chemicals and auxilaries
| Process | Chemicals | Brand/Origin |
| Dyeing of single jersey knitted fabric of organic cotton with reactive dye of Avitera Brand | Reactive dye (Avitera Brand) | Dycin |
| Gluber salt (Na2SO4.10H2O) | India | |
| Soda ash (Na2CO3) | India | |
| Soap | India | |
| Dyeing of single jersey knitted fabric of conventional cotton with reactive dye of Remazol Brand | Reactive dye (Remazol Brand) | Huntsman/Switzerland |
| Gluber salt (Na2SO4.10H2O) | India | |
| Soda ash (Na2CO3) | India | |
| Sequestering agent | Resotex-600S, India | |
| Wetting agent | Resotex-JSSA, India | |
| Leveling agent | Dyapol XLF, Yorkshire | |
| Soap | India |
Table 3.3: List of ecological test chemicals
| Process | Chemicals |
| Alkalinity test | Phenolphthalein indicator |
| Standard 0.02 N H2SO4 | |
| Methyl orange indicator | |
| COD | K2Cr2O7 |
| H2SO4 | |
| Ferrous ammonium sulphate | |
| Distilled Water | |
| BOD | Aerobic bacteria |
Table 3.4: List of physical test chemicals
| Process | Chemicals | Brand/Origin |
| Light fastness to perspiration | L-Histidine mono hydrochloride monohydrate (C6H9O2HClH2O) | James H.Heal, England |
| Sodium Chloride (NaCl) | James H.Heal, England | |
| Disodium hydrogen orthophosphate dehydrate (Na2HPO4.2H2O) | James H.Heal, England | |
| Color fastness to wash | HEAL’S (ISO) Standard Soap | James H. Heal, England |
| Anhydrous Sodium Carbonate | James H. Heal, England | |
| Absorbency test | Congo red | Germany |
3.3 Equipment
3.3.1 Equipment for Pretreatment and dyeing
- Dyeing vessel (Capacity 10 Liter)
- Thermometer
- Stirrer
- Graduated Cylinder
- Beaker
- Pipette
3.3.2 Equipment for Effluent test
- COD Reactor
- Micropipette
- Beaker
- UV Spectrophotometer
- Funnel
- Cylinder
- Filter paper
- Multi meter


Where,
A=Micropipette, B=COD vial, C=COD reactor, D=Spectrophotometer, E=Multi meter
3.3.3 Equipment for Physical test
Fabric drapability test
- Machine name: Fabric drape tester
- Model: M213
- Brand: SDL ATLAS
- Origin: UK

Bursting strength test
- Machine name: Universal strength tester
- Model: M250-3CT
- Origin: USA
Color fastness testing
Grey scale
Color change grey scale
- Brand: James H. Heal & Co. Ltd.
- Origin: England
- Test method: ISO-105-A02:1993

Color staining grey scale
- Brand: James H. Heal & Co. Ltd.
- Origin: England
- Test method: ISO-105-A03:1993

Light box
- Model: CAC-60
- Brand: Verivide
- Origin: UK

Multi fiber strip
- Brand: James H. Heal & Co. Ltd.
- Origin: England
Light fastness to rubbing
- Machine name: Crockmeter
- Company: James H.Heal & Company
- Origin: UK
- Model :670 Hand Driven CROCKMASTER

Light fastness to perspiration
- Machine name: Perspirometer
- Model: HX30
- Company: James H. Heal & Company
- Origin: UK

Machine name: Combined laboratory oven dryer
- Model: HX30
- Brand: James H. Heal & Company
- Origin: UK
Light fastness to wash
- Machine name: Gyrowash
- Model: 415/8
- Company: James H. Heal & Company
- Origin: UK

3.4 Manufacturing Methodology
3.4.1 Bio scouring of single jersey knitted fabric of organic cotton
Recipe
- Prima Green Eco scour: 0.20%
- Prima Fast Gold RSL: 0.50%
- Detergent: 0.30%
- Wetting agent: 0.20%
- M: L: 1:20
- Temperature: 60ºC
- Time: 60 min
- pH: 6
Working procedure

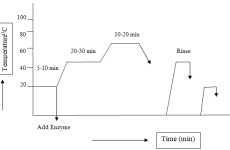
At first required amount of water according to recipe was taken in a vessel. Then required amount of detergent, wetting agent, Prima Green Eco Scour and Prima Fast Gold RSL were added to the dyeing vessel. Then pH of the solution was checked. After that fabric was added to the solution and treated for five minutes at normal temperature. Then the temperature was raised to 60ºC and the treatment was carried out for 60 minutes. After that the temperature was raised to 80ºC and the treatment was carried for 10 minutes and then the bath was dropped. Then hot wash of the sample was done using required amount of detergent at 80ºC temperatures for five minutes.
3.4.2 Scouring of single jersey knitted fabric of conventional cotton
Recipe
- NaOH: 5 g/L
- Wetting agent: 1 g/L
- Detergent: 2 g/L
- Sequestering agent: 1.5 g/L
- M: L: 1:25
- Temperature: 95ºC-100⁰C
- Time: 45 min
- pH: 10.5-11.0
Working procedure

At first required amount of water according to recipe was taken in a vessel. Then required amount of detergent, wetting agent, sequestering agent and NaOH were added to the vessel. Then pH of the solution was checked. After that fabric was added to the solution and treated for five minutes at normal temperature. Then the temperature was raised to 95ºC and the treatment was carried out for 45 minutes and the bath was dropped. After that neutralization was done using required amount of acetic acid at normal temperature for five minutes. Then hot wash of the sample was done using required amount of detergent at 80ºC temperature for ten minutes.
3.4.3 Dyeing of single jersey knitted fabric (organic cotton) with reactive dye of Avitera brand
Recipe
- Dyestuff: 3%
- Gluber salt: 50 g/L
- Soda ash: 15 g/L
- Temperature: 60ºC
- Time: 45 min
- pH: 10-11
- M: L: 1: 25
Working procedure

At first required amount of water according to recipe was taken in a vessel. Then required amount of salt and dye were added to the vessel. Then pH of the solution was checked. After that fabric was added to the dye bath and treated for five minutes at normal temperature. Then raising the temperature up to 60ºC half of total amount of soda was added and the treatment was carried out for 15 minutes. Then the rest amount of soda was added and the treatment was carried out for 30minutes.Then the bath was dropped. After that hot wash at 80ºC for 10 minutes and cold wash at room temperature were done sequentially.
3.4.4 Dyeing of single jersey knitted fabric (conventional cotton) with Reactive dye of Remazol Brand
Recipe
- Dyestuff: 3%
- Salt: 50 g/L
- Soda ash: 15 g/L
- Sequestering agent: 1.5 g/L
- Wetting agent: 1.5 g/L
- Leveling agent: 1.5 g/L
- Temperature: 60ºC
- Time: 45 min
- pH: 10-11
- M: L: 1:20
Working procedure

At first required amount of water according to recipe was taken in a vessel. Then required amount of salt, sequestering agent, wetting agent, leveling agent and dye were added to the vessel. Then pH of the solution was checked. After that fabric was added to the dye bath and treated for five minutes at normal temperature. Then raising the temperature up to 60ºC half of total amount of soda was added and the treatment was carried out for 15 minutes. Then the rest amount of soda was added and the treatment was carried out for 30minutes.Then the bath was dropped. After that hot wash at 80ºC for 10 minutes and cold wash at room temperature were done sequentially.
3.4.5 Apparel manufacturing
Measurement
- Body length: 68 cm
- ½ Chest: 43 cm
- Sleeve length: 20cm
- Arm hole circumference: 46cm
- Neck circumference: 44cm
- Back neck drop: 1cm
- Front neck drop: 8cm
Process sequence

List of machine and materials
- Pattern paper
- Scissors
- Sewing thread
- Four thread overlock machine
- Single needle lock stitch machine
- Flat lock machine
Operation sequence of sewing
Table 3.5: Sewing operation sequence of T-shirt
| Sl.No. | Operation | Machine |
| 01 | Shoulder join | OL4 |
| 02 | Rib tuck | LS1 |
| 03 | Rib join | OL4 |
| 04 | Back tape join | LS1 |
| 05 | Piping topsin | FL |
| 06 | Rib front topsin | FL |
| 07 | Sleeve join | OL4 |
| 08 | Side seam join | OL4 |
| 09 | Sleeve tuck | LS1 |
| 10 | Body hem | LS1 |
3.4.6 Printing process

Process sequence

Screen preparation
Raw materials
- Screen frame
- Screen gauge
- Glass plate
- Chrome gelatin solution
Recipe for preparing sensitive solution
Solution A:
- Pure gelatin =200gm
- Hot water =500gm
- Total =700gm
Solution B:
- Ammonium dichromate =70gm
- Hot water =150gm
- Liquor ammonia =80gm
- Total =300gm
By mixing solution, A and B in room temperature 1000gm sensitive solution was made.
Working procedure
To prepare the screen for printing photo chemical method was followed. At first the screen was treated with chrome gelatin and dried at room temperature to make the screen sensitive. A positive design was made on tracing paper by using opaque ink. Then the positive design was attached with the screen and the screen was placed in glass plate. After that light was passed through the glass plate. As light passed through the white portion of the positive design, gelatin of this portion became hard. On the other hand, gelatin of opaque portion of the positive design remained soluble as light did not pass through this portion. Then removing the glass plate, the screen was washed in hot water and then in cold water to remove the soluble gelatin from the screen.
Printing of knitted fabric (Organic cotton) with reactive dye (Avitera brand)
Stock thickener preparation procedure
- CMC thickener = 5 parts
- Water = 95 parts
- Total = 100 parts
CMC was added into water while stirring and it was kept stirring for at least 20 minutes.
Preparation of print paste
- Reactive dye (Avitera Brand) = 5 parts
- Urea = 8 Parts
- Resist salt = 1 Parts
- Glycerin = 2 Parts
Then it was bulked to 100 parts by adding the thickener. Then paste was heated for 5 minutes and finally the paste was ready for printing.
Printing of knitted fabric (Conventional cotton) with reactive dye (Remazol brand)
Stock thickener preparation procedure
- CMC thickener = 5 parts
- Water = 95 parts
- Total = 100 parts
CMC was added into water while stirring and it was kept stirring for at least 20 minutes.
Preparation of print paste
- Reactive dye (Remazol Brand) = 5 parts
- Urea = 8 Parts
- Resist salt = 1 Parts
- Glycerin = 2 Parts
Then it was bulked to 100 parts by adding the thickener. Then paste was heated for 5 minutes and finally the past was ready for printing.
Printing procedure
At first the sample was placed on the printing table and the screen was placed on the sample. After that print paste was poured on the screen and the paste was squeezed by using a squeezer. Then the sample was steamed at 102ºC for 2 minutes and curing was done at 130ºC for five minutes.
3.4.7 Hang tag
Normally necessary information like brand name, origin, product type is mentioned in a hang tag. Besides this some information to make aware the customer can be included in hang tag. A typical hang tag has been developed through which some information regarding environmental consciousness has been conveyed.

3.5 Effluents testing procedure
3.5.1 Determination of DO, Conductivity and pH
Apparatus
- Multimeter
- pH Port
- DO Port
- Conductivity port
- Beaker
- Waste water
Testing procedure

Waste water is taken in a beaker. pH port, DO port and Conductivity port were set to multi meter for determining pH, DO and Conductivity respectively. Then the port was dipped into the waste water and kept for few seconds until the result was shown in the display. That pH, DO (mg/L) and conductivity (mS/cm) values were found directly from multi meter.
3.5.2 Determination of Alkalinity
The alkalinity of water is a measure of its capacity to neutralize acids. Alkalinity is measured volumetrically by titration with N/50 (i.e. 0.02 N) H2SO4 and is reported in terms of equivalent CaCO3.
Reagents
- Phenolphthalein indicator
- Standard 0.02 N H2SO4
- Methyl orange indicator
Apparatus
- Beaker
- Measuring cylinder
- Burette pillar
- Magnetic Stirrer
Testing procedure
- At first 100ml of the sample is taken into a beaker.
- Then pH of the sample is determined.
- After that 3 drops of Phenolphthalein indicator are added to the sample. If the sample becomes pink then it is to follow step 4 otherwise to follow step 5.
- 0.02 N H2SO4 from a burette is to be added to the sample until the pink color just disappears. It is to record the ml. of the acid used.
- 3 drops of Methyl orange indicator are to be added to the sample. If the sample becomes yellow and 0.02 N H2SO4 acid until the first change in color is noted. The end point is slight orange tinge. It is to record the ml. of the acid used.

Where,
A=200 times diluted dyeing effluents, B=Phenolphthalein indicator added solⁿ, C=0.02 N H2SO4 added solⁿ.
Calculation procedure

3.5.3 Measurement of BOD
BOD is the amount of oxygen used by aerobic bacteria to decompose organic material. This process may take place many days hence to standardize the measurement of BOD following standards are taken
- Time (Incubation period): 5 days
- Temperature: 20ºC
Apparatus
- BOD Oxitop
- BOD Incubator
- Waste water
- Distilled water

Working procedure
- Pure distilled water saturated with oxygen was taken in BOD Oxitop.
- 5ml of polluted water+25ml of pure water was taken in another BOD Oxitop.
- Then the both oxitop was kept for five days in BOD incubator at 20ºC.
- Then dissolved oxygen of pure water and dilute impure water were measured after 5 days.
- BOD was found from the difference in dissolved oxygen of two reading.
3.5.4 Measurement of COD
Any kind of chemically oxidizable impurities present in effluent is COD. It is the measure of both biologically oxidizable and biologically inert organic matter.
Apparatus
- COD Reactor
- Micro pipette
- Beaker
- COD Vial
- UV Spectrophotometer

Working procedure
First effluents were taken in COD vial. Then the vial was kept in COD reactor at 150⁰C for 2 hours. Then the vial was put off from COD reactor and cooled in normal temperature. After that the sample was tested in UV spectrophotometer with respect to the fresh COD vial. Before testing the UV spectrophotometer was calibrated carefully. COD value in mg/L of the sample was found directly from UV spectrophotometer.
3.5.5 Determination of total solid dissolved solid and suspended solid
Total solid (TS) refers to the matter that remains as residue upon evaporation and drying at 103-105⁰C.The total solid includes total suspended solid (TSS)-the portion of the total solid retained by filter and total dissolved solid (TDS)-the portion that passes through the filter.
Apparatus
- Beaker
- Measuring cylinder
- Funnel
- Dropper
- Filter paper
- Oven
- Electric balance

Testing procedure
Total solids
- A clean glass beaker was taken (which was kept at 103ºC in an oven for 1 hour) of 150 ml capacity and also the weight of the beaker was taken.
- Then 100 ml of the sample was poured into the beaker.
- After that the beaker was placed in an oven maintained at 103ºC for 24 hours. Then cooling the beaker weight of the beaker was taken. Weight of the solid was found by subtracting this value from the weight of clean beaker.
Calculation
Total solids, TS (mg/l) = (mg of solids in the beaker×1000)/volume of sample
Dissolved solid
- A clean glass beaker was taken (which was kept at 103ºC in an oven for 1 hour) of 150 ml capacity and also the weight of the beaker was taken.
- 100 ml of the sample was taken and filtered it through a double layered filter paper and the filtrate was taken in a beaker.
- After that the beaker was placed in an oven maintained at 103ºC for 24 hours. Then cooling the beaker weight of the beaker was taken. Weight of the solid was found by subtracting this value from the weight of clean beaker.
Calculation
TDS (mg/l) = (mg of solids in the beaker×1000)/volume of sample
Suspended solid
Total suspended solid, TSS (mg/Lt) = TS (mg/L) – TDS (mg/L)
3.6 Physical Properties testing procedure
3.6.1 Color fastness to wash
Recipe
- Soda ash (Na2CO3): 2 g/L
- ISO standard soap: 5 g/L
- M: L: 1:50
- Time: 30
- Temperature: 60ºC
- Test method: ISO-105-CO3
Apparatus and materials
- Gyrowash machine (Wash wheel)
- Sample size (10cm×4cm)
- Multi fiber fabric (5cm×4 cm)
- Sewing machine
- Color change grey scale
- Color staining grey scale
- Light box
Working procedure
A sample was cut. Then a multifiber fabric was attached to it by sewing. A solution was made according to the recipe mentioned above. The solution was taken into wash wheel. Then the sample was dipped into it. After that the wash wheel was set into Gyrowash machine and the machine was run for 30 minutes at 60ºC temperature. Then the sample was put off from the machine and rinsing, squeezing, removing of stitching, drying (in oven) were done sequentially. Color fastness of the sample i.e. color fading and color bleeding was checked using color change grey scale and color staining grey scale respectively. Both the evaluation was done under standard light using light box.
3.6.2 Color fastness to perspiration
Recipe
- Histidine Monohydrochloride monohydrate (C6H9O2HClH2O): 0.5 g
- Sodium Chloride (NaCl): 5g
- Disodium hydrogen orthophosphate dehydrate (Na2HPO42H2O): 2.5 g
Apparatus and materials
- Perspirometer
- Oven
- Multifiber fabric
- Grey scale
- Sample size (10cm×4cm)
- Multi fiber fabric (5cm×4 cm)
- Test method: ISO-105-E04-1994(E)
Working procedure
A sample was cut. Then a multi-fiber fabric was attached to it by sewing. A solution of 1000 ml was made by following the recipe mentioned above. Then the composite test sample dipped into the solution and kept for 30 minutes. Then the sample was taken off from the solution and was put into two glass plates of perspirometer. After that 12.5Kpa pressure was inserted onto the glass plates. Then the perspirometer wash kept in an oven for four hours at 37ºC temperature. Then the sample was put off from the oven. Color fastness of the sample i.e. color fading and color bleeding was assessed using color change grey scale and color staining grey scale respectively. Both the evaluation was done under standard light using light box.
3.6.3 Color fastness to rubbing
Apparatus and materials
- Crockmeter
- Sample size (15cm×4cm)
- Rubbing cloth size (4cm×4 cm)
Color staining grey scale
- Light box
- Distilled water
- Test method: AATCC Test method 8-2007
Working procedure
A sample having the dimension of 15cm×4cm was cut and then set in crockmeter. Then a rubbing cloth was fastened to the weighted finger. The finger was permitted to rub the sample 10 times by driving the handle. The work was done for wales direction. For wet rubbing the center portion of the rubbing cloth was wetted. The rubbing cloth wash compared with fresh rubbing cloth using color staining grey scale.
3.6.4 Fabric drapability test
Drape is the term used to describe the way a fabric hangs under its own weight. Fabric drapability is an important factor from an aesthetic point of view. The drape co-efficient has been developed to describe the degree of drape and drape shade. In actual test the light beam casts a shadow of the draped fabric onto a ring of highly uniformed translucent paper supported on a glass screen. The surface of drape pattern area on the paper ring is directly proportional to the mass of that area, so the drape co-efficient (F) can be calculated in the simple way.

A lower F value means the fabric is softer and its drapability is better. In other word, the higher the drape co-efficient, the stiffer the fabric is.
Apparatus and materials
- Fabric drape tester
- Translucent paper ring
- Template (Diameter=18cm)
- Fabric sample
- Electric balance
Working procedure
In the test a circular specimen was held concentrically between two smaller horizontal discs and was allowed to drape into folds under its weight. A light was shown from underneath the specimen and the shadow that the fabric cast was traced onto an annular piece of paper, the same size as the unsupported part of the fabric specimen. The whole was weighted and then the shadow part of the ring was cut away weighted. Finally, by using the formula mentioned above drape co-efficient was determined to assess the draping properties of the fabric.
3.6.5 Bursting strength test
Testing parameters
- Machine name: Universal Strength tester
- Test name: Ball burst
- Test speed: 305 mm/min
- Preload: 0.100 lb.
- Diameter: 1.75 inches
- Sample size: 5”×5”
- Gauge length: 320cm(upper),220cm(lower)
- Clamp dia: 4.5cm
- Test method: ASTM-D-3787
Working procedure
At first ball burst attachment was set, required parameters for the test were set and also the machine was calibrated. Then the sample was set in the clamp. After that the machine was run. When the sample was burst due to the force applied by the attachment, the machine was automatically stopped and the machine gave direct reading of bursting strength of the sample. Same procedure was done for different sample and the result was taken.
3.6.6 Absorbency test
Drop test
In a pipette a solution of 0.1% direct red was taken and was dropped on the fabric on the fabric sample from 5cm above. Then the absorption of the color drop was observed visually and time in second to absorb one drop of solution was taken.
Spot test
In a pipette a solution of 0.1% direct red was taken and droplet of solution was put on the different places of the fabric. Then the shape of the absorbed area on the fabric was observed.

Where,
1 = Fabric of conventional cotton treated with NaOH
2 = Fabric of organic cotton treated with enzyme
4. RESULTS AND DISCUSSION
4.1 Ecological test results
Table 4.1: Test results of ecological parameters
| Sample No. | Source of Effluents | Effluents content | Test name | Results | DoE Standard |
| Sample-1 | Scouring | Wetting agent, Detergent, Sequestering agent, Caustic soda, Cotton waste | BOD in mg/L | 1500 | 50 |
| COD in mg/L | 2700 | 200 | |||
| DO in mg/L | 1.06 | 6 | |||
| EC in mS/cm | 32 | 12 | |||
| TDS in mg/L | 16120 | 2100 | |||
| TSS in mg/L | 258 | 150 | |||
| ALKANITY in mg/L | 800 | 500 | |||
| pH | 12.69 | 7 | |||
| Sample-2 | Enzymatic scouring | Detergent, Wetting agent, Prima green Eco scour enzyme, Prima fast Gold RSL enzyme | BOD in mg/L | 1000 | 50 |
| COD in mg/L | 2200 | 200 | |||
| DO in mg/L | 2.76 | 6 | |||
| EC mS/cm | 6.27 | 12 | |||
| TDS in mg/L | 3130 | 2100 | |||
| TSS in mg/L | 151 | 150 | |||
| ALKANITY in mg/L | 700 | 500 | |||
| pH | 6.41 | 7 | |||
| Sample-3 | Dyeing | Reactive dye (Remazol brand), Sodium chloride, Sodium carbonate, Sequestering agent, Wetting agent, Leveling agent | BOD in mg/L | 800 | 50 |
| COD in mg/L | 1989 | 200 | |||
| DO in mg/L | 2.23 | 6 | |||
| EC mS/cm | 59.1 | 12 | |||
| TDS in mg/L | 29500 | 2100 | |||
| TSS in mg/L | 585 | 150 | |||
| ALKANITY in mg/L | 1200 | 500 | |||
| pH | 11.27 | 7 | |||
| Sample-4 | Dyeing | Reactive dye (Avitera brand), Sodium chloride, Sodium carbonate | BOD in mg/L | 200 | 50 |
| COD in mg/L | 644 | 200 | |||
| DO in mg/L | 2.92 | 6 | |||
| EC mS/cm | 57.1 | 12 | |||
| TDS in mg/L | 22700 | 2100 | |||
| TSS in mg/L | 430 | 150 | |||
| ALKANITY in mg/L | 800 | 500 | |||
| pH | 11.16 | 7 |
4.1.1 Bio-chemical Oxygen Demand (BOD)

The BOD (mg/L) values of the effluents for organic cotton after bio scouring is a greater extent lower than the conventional cotton caustic scouring. This is because the use of enzyme in place of caustic soda and maintaining a lower temperature around 60⁰ c. And the BOD (mg/L) value of the effluents for organic cotton scoured fabric after dyeing with low impact reactive dyes it shows a better and lower value i.e. 200 mg/L which is near about the DOE standard BOD value 50 mg/L. On the other hand, the conventional cotton scoured knit fabric after treated with cold brand Remazol reactive dyes the BOD value of the effluents increases to 800 mg/L. Hence, it can be concluded that the scouring of organic cotton knit fabric with the enzyme can reduce the BOD of the effluents to a considerable extent as compared to that of scouring with the caustic soda of conventional cotton knit fabric.
4.1.2 Chemical Oxygen Demand (COD)

The COD (mg/L) values of the effluents for organic cotton after bio scouring and low impact dyeing and conventional cotton after scouring and dyeing are presented in Figure 4.2. The results show that the COD of the effluents does not show much difference between the organic cotton bio scouring (2200 mg/L) and conventional cotton scouring (2700 mg/L). Sample 2 is just a bit lower than the sample 1. But the COD value of the effluents of organic cotton knit dyed fabric by low impact reactive dye shows a greater reduction than conventional cotton reactive dye effluents. The COD value is 644 mg/L for organic cotton low impact dye effluents which is adjacent to the DOE standard COD value 200 mg/L. Hence, it can be concluded that the dyeing of fabric with the low impact dyes can reduce the COD of the effluents to a considerable extent as compared to that of dyeing with the regular dyes using cold brand reactive dye.
4.1.3 Dissolved Oxygen (DO)

The DO (mg/L) values of the effluents for organic cotton after bio scouring with low impact dyeing and conventional cotton after scouring and conventional dyeing are presented in Figure 4.3. The results show that the DO value in mg/L of the effluents collected after bio scouring of organic cotton shows a greater value 2.76 mg/L in comparison of conventional cotton scouring effluents only 1.06 mg/L. And in case of low impact reactive dyeing it again shows a greater DO value 2.92 mg/L than the conventional cotton knit fabric reactive dyeing. For both cases the DOE standard of DO value is 6 mg/L. DO value shows the amount of oxygen dissolved in the soil or bodies of water. Environmental oxygenation can be important to the sustainability of a particular ecosystem. So, the more they DO in water the better for environment.
4.1.4 Electrical Conductivity (EC)
Effluents collected from pretreatment and dyeing stages were tested by using Multimeter to measure EC or conductivity. This test was done to know the purification level of the effluents with respect to normal water. It can be noted that lower the EC of any liquid, the more pure the liquid is.

EC of organic sample was well satisfactory as there was no electrolyte in the effluents so its purification was also satisfactory. On the other hand, conventional sample showed less purity as its EC value is so much higher than the normal water. Both the effluents collected from dyeing process i.e. from conventional and organic showed higher value of EC as chemical and auxiliaries used in dyeing worked as electrolyte solution.
4.1.5 Total Suspended Solid (TSS)
As TSS refers to the amount of insoluble matter in liquid so higher TSS value of effluents discharged into water can reduce the quality of water.

Organic sample showed excellent result as it was collected from enzymatic scouring. On the other hand, conventional scoured sample showed higher TSS value. In dyeing reactive dye was used so the effluents collected from dyeing process shows higher value of TSS. However organic sample shows lower TSS value as compared with conventional cotton knit fabric. This is because of the application of low impact reactive dye on organic cotton knit fabric.
4.1.6 Total Dissolve Solid (TDS)
Higher TDS of effluents discharged into water tends to reduce the quality of water. TDS of effluents collected from scouring and dyeing process were tested for both type of fabric.

TDS result of sample 2 is well satisfactory as compared with sample 1. This is because enzyme was used for scouring process. Caustic and other inorganic materials were used in conventional scouring so sample 1 shows higher TDS. Both the Effluents collected from dyeing process show higher TDS because of the hydrolyzed reactive dyes and a huge amount of salt and soda content. But TDS of sample 4 shows less TDS value than sample 3 because of the application of low impact reactive dye.
4.1.7 pH
pH of effluents collected from of pretreatment and dyeing stages were tested by using Multimeter. This test was done to know the pH level of the effluents as more acidity or alkalinity of effluents that are discharged into water are not good for maintaining a balanced ecosystem.

pH level of the effluents collected from enzymatic scouring of organic cotton fabric was well satisfactory as it was near to 7. This was because here enzyme i.e. microorganism was used as scouring chemical. On the other hand, pH of the effluents of conventional cotton fabric scouring with NaOH was highly alkaline which is not safe to discharge into water. Effluents collected from dyeing process of both samples showed alkaline pH in dyeing stage because the effluents contain gluber salt and soda ash.
4.1.8 Alkalinity
Alkalinity is the name given to the quantitative capacity of an aqueous solution to neutralize an acid.

In sample 1 the alkalinity shows greater value because of using caustic soda during scouring. On the other hand, using enzyme reduces the alkalinity of the effluents in sample 2. In dyeing both in sample 3 and sample 4 the effluents contains a great amount of soda ash and gluber salt which increases the alkalinity of the effluents. The DOE standard of alkalinity is 500 mg/L. Sample 2 and sample 4 shows some adjacent result to the standard than sample 1 and sample 3.
4.2 Physical testing results
4.2.1 Estimation of scouring effect
Results of drop test
Table 4.2: Drop test result
| Sample description | Absorption time in second |
| Knitted fabric of organic cotton treated with Enzyme | 0.47 |
| Knitted fabric of conventional cotton treated with Caustic soda | 1.02 |
Knitted fabric of organic cotton treated with Enzyme shows excellent scouring effect than the fabric sample of conventional cotton as it took lower time to absorb one drop of color solution.

Results of spot test
Spot test of the sample shows that scouring effect of the fabric sample of organic cotton is more uniform than the sample of conventional cotton.

Where,
1= Fabric of conventional cotton treated with NaOH
2=Fabric of organic cotton treated with enzyme
4.2.2 Results of fabric weight loss due to scouring
Table 4.3: Fabric weight loss assessment result
| Weight before scouring (gm.) | Weight after scouring (gm.) | Weight loss% | |||
| Sample of org. cotton | Sample of conv. cotton | Sample of org.cotton | Sample of conv. cotton | Sample of org. cotton | Sample of conv. cotton |
| 350 | 310 | 318 | 294 | 9.14 | 5.16 |
It has been noticed that weight loss% of organic cotton sample is comparatively higher than the conventional cotton sample.

4.2.3 Results of Color fastness to Wash
Table 4.4: Color fastness to Wash assessment result
| Test result in grade for knitted fabric of organic cotton | Test result in grade for knitted fabric of conventional cotton | |
| Color change | 4/5 | 4/5 |
| Color staining | ||
| Di-acetate | 4/5 | 4/5 |
| Cotton | 4/5 | 4/5 |
| Nylon | 4/5 | 4 |
| Polyester | 4/5 | 4/5 |
| Acrylic | 4/5 | 4/5 |
| Wool | 5 | 4/5 |

Slight difference in the color fastness to wash of the both sample was found.
4.2.4 Test result of color fastness to rubbing
Table 4.5: Color fastness to rubbing assessment result
| Sample Type | Test result in grade | |
| Dry | Wet | |
| Knitted fabric sample of organic cotton | 4/5 | 4 |
| Knitted fabric sample of convention cotton | 4/5 | 3/4 |

Though result of dry rubbing was same for the both samples, slight difference was found in wet rubbing.
4.2.5 Test results of color fastness to perspiration
Table 4.6: Color fastness to perspiration assessment result
| Test result in grade for knitted fabric of organic cotton | Test result in grade for knitted fabric of conventional cotton | |
| Color change | 4/5 | 4/5 |
| Color staining | ||
| Di-acetate | 5 | 5 |
| Cotton | 4/5 | 4 |
| Nylon | 4/5 | 4/5 |
| Polyester | 4/5 | 4/5 |
| Acrylic | 4/5 | 4/5 |
| Wool | 4/5 | 4/5 |

No noticeable difference was found in the result of color fastness to perspiration for both the sample of organic cotton and conventional cotton.
4.2.6 Test results for Ball Bursting Strength
Table 4.7: Ball Bursting Strength test results
| Ball Bursting Strength (N) | |||
| Knitted fabric sample of organic cotton | Knitted fabric sample of conventional cotton | ||
| Grey | Dyed | Grey | Dyed |
| 291.560 | 236.580 | 288.290 | 326.600 |
It was noticed that ball bursting strength for fabric of organic cotton was fall due to enzymatic scouring whereas strength of fabric sample of conventional cotton was increased due to scouring using caustic soda.

4.2.7 Result for fabric Drape test
Table 4.8: Fabric Drape test result
| Sample Type | Drape co-efficient | Drapability |
| Sample of organic cotton | 64.94 | Excellent |
| Sample of conventional cotton | 76.97 | Good |

Result of drape test was well satisfactory for organic cotton sample as it shows a lower value of drape co-efficient that means the fabric is softer and easily drapable than the sample of conventional cotton.
4.3 Hang Tag
A hang tag has been attached to the developed green apparel product that conveys messages to the consumers to make them aware about green product. As the hang tag contains maximum information transparency so the customers can be easily acquainted with the characteristics of the green apparel product.

4.4 Developed apparel product
Two green apparel products have been developed using separate raw materials. Each product shows their respective properties. The organic cotton sample is more environment friendly as it contains organic cotton and in processing stage no hazardous chemicals have been used. So, it can be referred to as green apparel product. On the other hand, conventional cotton T-shirt is not environment friendly as it contains conventional cotton and in processing stage hazardous chemicals have been used.


4.5 Results of RDM (Rapid Design Module)
The RDM-Beta is a prototype designed to test the concept of a simple tool that aims at educating and providing quick directional guidance to apparel and footwear designers during the product creation process about the potential environmental impacts of their design solutions.
4.5.1 For organic cotton knit T-shirt


4.5.2 For Conventional Cotton Knit T-shirt


Discussion of Higg index assessment
The Higg Index 2.0 is a suite of sustainability assessment tools that anyone can get started with right away. These assessments, called modules, evaluate impacts through three different lenses: Facility, Brand, and Product. The Higg Index 2.0 is primarily an indicator-based assessment tool for apparel and footwear products. The Index asks practice-based, qualitative questions to gauge environmental sustainability performance and drive behavior for improvement. The RDM-Beta in not a substitute for the Produce Module but is a product-focused tool in the Higg Index 2.0 suite of tools.
In this module five steps were focused. They are:
- Material.
- Manufacturing.
- Packaging.
- Product Use.
- End Use.
For organic t-shirt all the inputs for the five types of data indicate environment sustainability. Such as the material used 100% organic, low impact dyeing method is used, basic rinse finish is used, sustainable hang tag with sustainable care instruction is included, medium impact care is needed during the products use and finally the reuse of the material is possible. As a result, the RDM assessment shows a good score 63 out of 100.
On the other hand, for conventional cotton knit t-shirt the material is not 100% organic, the marker efficiency is 85%, traditional dyeing method is used, basic rinse finish is performed, no packaging or care instruction is included. Under these circumstances the RDM assessment tool shows a poor score 37 out of 100.
5. CONCLUSION
5.1 Conclusion
This thesis mainly focuses on eco-friendly product development from material selection to end product. Firstly, the selection of organic cotton over conventional cotton ensures the journey towards eco-friendly product development. In the subsequent processes the selection of enzymatic scouring, low impact reactive dyes for dyeing and printing and small printing area also ensures the low environmental impact than conventional cotton fabric processing to product development. The results shows that the use of organic fabric, enzyme and low impact dye has a greater ecological advances as it reduces the BOD, COD, TDS, TSS, Alkalinity and Pᴴ of the effluent and increases the DO value in both scouring and dyeing stages than the conventional cotton, caustic scouring and cold brand reactive dyeing processes. The bleaching process is avoided here by dyeing in deep shade which also reduces the amount of toxic effluent. The selected physical properties test results also show similar or better result of organic cotton knit fabric with conventional cotton knit fabric except the lower bursting strength. Finally, printing has been done by using low impact reactive dye in a small printing area which is also a symbol of eco-friendly. A hang tag has been developed which provides required care information to the consumer and the most important aim of the hang tag is to provide the information that can aware the consumer to think green and be sustainable. Reuse and recycle information of the product has been included in the hangtag.
5.2 Limitations
This thesis work has been done on only one shade of dye. The effluents of dyeing by different shades of dye can be tested. Due to the insufficiency of monetary support and time the toxicity, carcinogen, azo, formaldehyde and other metal content tests of the apparel cannot be performed. If natural thickener for printing and organic cotton sewing thread were used then the apparel would be more environment friendly. Due to some lack of information the costing for both type of apparel cannot be measured.
5.3 Future Scope
- The environmental impact of different shades of low impact reactive dyes can be measured.
- The costing for both type of apparel can be determined and a comparative study can be done.
- It can be determined how to initiate bulk production of environment friendly apparel product.
- Several attempts to increase customer consciousness about environment friendly apparel can be done.
6. REFERENCES
- SHERBURNE, (2011). Achieving Sustainable Textiles: A designer’s perspective.
- Chouinard, Y., & Brown, M. S. (1997). Going Organic: Converting Patagonia’s Cotton Product Line. Journal of Industrial Ecology, 1(1), 117–129. doi:10.1162/jiec.1997.1.1.117
- HAE Now Organic Tee. (2013). Retrieved from HAE Now Organic Tee Web site: http://www.haenow.com/cart/whyorganic.php
- Mihelcic, J.R., Crtittenden, J.C., Small, M.J., Sonnard, D.R., Hokanson, D.R., Zhang, Q., Chen, H., Sorby, S.A., James, V.U., Sutherland, J.W. and Schooner, J.L. (2003) Sustainability Science and Engineering: Emergence of a New Metadiscipline. Environmental Science and Technology, 37(23) 5314-5324.
- Murugesh Babu K & Selvadass M, (2012). Investigation on Ecological Parameters of Dyeing Organic Cotton Knitted Fabrics. Universal Journal of Environmental Research and Technology, Volume 2, Issue 5: 421-428
- Noor, S. S. & Rohasliney, H. (2011): A Preliminary Study on Batik Effluent in Kelantan State: A Water Quality Perspective. International Conference on Chemical, Biological and Environment Sciences, Bangkok, 274-276.
- R.S.Blackburn,(2009). Sustainable Textiles Life Cycle and environmental impact.UK:Woodhead Publications.
- Sustainable clothing From Wikipedia, the free encyclopedia (last modified on 28 April 2014). Retrieved from http://en.wikipedia.org/wiki/Sustainable_clothing
- Sustainable Apparel Coalition. (2012). Retrieved from Sustainable Apparel Coalition: http://www.apparelcoalition.org/rapid-design-module-rdm-beta/
- Theodore Roszak. (2001). The Voice of the Earth: An Exploration of Ecopsychology. Phanes Press.
- Tomasino, D. C. (1992). Chemistry & Technology of Fabric Preparation & Finishing. North Carolina: Department of Textile Engineering, Chemistry & Science College of Textiles North Carolina State University.
- Wikipedia. (2014, june 2). Retrieved from Wikipedia: http://en.wikipedia.org/wiki/Biochemical_oxygen_demand
- Wikipedia. (2014, june 2). Retrieved from Wikipedia: http://en.wikipedia.org/wiki/Biochemical_oxygen_demand
- Wikipedia. (2014, may 18). Retrieved from Wikipedia: http://en.wikipedia.org/wiki/Oxygen_saturation
- Wikipedia. (2014, may). Retrieved from Wikipedia: http://en.wikipedia.org/wiki/Alkalinity
- Wikipedia. (2014, may). Retrieved from Wikipedia: http://en.wikipedia.org/wiki/Total_dissolved_solids
- Wikipedia. (2014, may). Retrieved from Wikipedia: http://en.wikipedia.org/wiki/PH
Founder & Editor of Textile Learner. He is a Textile Consultant, Blogger & Entrepreneur. Mr. Kiron is working as a textile consultant in several local and international companies. He is also a contributor of Wikipedia.