Estimation of Scouring Effect on Cotton by Varying Concentration of Caustic Soda
Chaitanna Sarkar & Muhammad Abdur Rashid
Department of Textile Engineering
Dhaka University of Engineering & Technology (DUET)
Gazipur -1700, Bangladesh
ABSTRACT
Apparel knit fabrics are normally prepared and dyed in exhaust equipment such as winch dyeing machine and jets. Scouring, prior to bleaching and dyeing is usually done in an alkaline process by boiling cotton in sodium hydroxide. Alkaline scouring at high temperatures for a long period of time followed by bleaching can abrade the fabrics and create an undesirable damage and change on the fabric surface.
Scouring is practiced in the textile industry to remove the non-cellulosic hydrophobic cuticle constituents. This improves wettability of the fibers, which facilitates uniform dyeing and finishing. Conventionally, scouring is performed by hot hydrolysis with NaOH, which involves large quantities of water and energy and requires special handling of the strong alkaline effluents
This project & thesis work has been devoted to study the scouring effect specially the minimum weight loss percentage (%) and obtaining maximum absorbency (Drop test, Immersion test) of textile material using minimum caustic soda.
CHAPTER 1
INTRODUCTION
1.1 Introduction
Most of the research done to develop an innovative alkaline scouring process was focused on the potentials of different caustic concentration. In this study a rational approach is adopted to design a new efficient caustic based cotton scouring process. The main aim is to find suitable caustic for the scouring purpose and to use it efficiently in the process. To do so, detailed knowledge of cotton fiber structure is required. A question that needs to be answered for designing a new scouring process is, what to modify in cotton fibers to make them hydrophilic. Related questions are which specific components need to be removed and how to remove these unwanted components from the fiber. Apart from studying, each constituent in detail, the knowledge of how these unwanted cotton components are interconnected vital. Knowledge of the interactions between cotton fibers and caustic/chemicals/additives is essential for proper interpretation of the obtained results. All these issues are directly related to the fundamental knowledge about cotton fiber structure and morphology. A thorough literature review has been done on this issue, including the latest insights from plant science. That reveals structural information that directly and strongly influences the selection of caustic amount for developing a new scouring process. The gathered information about the cotton fiber is focused on those aspects that can affect the performance of an caustic scouring process. To complete the cotton fiber structure, some additional information on the secondary wall, coloring matters and the metal contents as well have been given.
1.2 Objectives of the project & thesis
- To ensure minimum weight loss (%) of cotton.
- To obtain maximum absorbency.
- To use minimum caustic soda for performing Scouring Process.
- To increase scouring efficiency
- To save process cost.
1.3 Cotton fiber
Cotton is a seed hair fiber of the plant belonging to Gossypium, the botanical genus of the mallow family. After flowering, an elongated capsule or boll is formed in which the cotton fibers grow. Once the fibers grow completely, the capsule bursts and fibers come out. A cotton capsule contains about 30 seeds and each seed hosts around 2000 to 7000 seed hairs (fibers). Depending on the cotton type and growing conditions, the color of the fiber is usually creamy white or yellowish. The average fiber length of different kinds of cotton varies from 22 to 50 mm, and its diameter, from 18 to 25 μm. The higher quality fibers are known as long-staple fibers or extra-long staple.
1.4 Cross section of cotton fiber
The cell wall is a dynamic structure which composition and form can change markedly, not only during cell growth but also after the cells have become matured . The cotton fiber is structurally built up into concentric zones and a hollow central core known as the lumen.

The mature fiber essentially consists of (from outside to inside) – the cuticle i.e. the outermost layer, the primary cell wall, the secondary wall and the lumen . Figure 1.1 systematically shows the different layers present in the cotton fiber with the compositions of each layer. Cotton contains nearly 90% of cellulose and around 10% of non-cellulosic substances, which are mainly located in the cuticle and primary wall of the fiber. Typical components in dry mature cotton fibers are given in Table 1.1. From this table it is clear that most of the non-cellulosic materials are present in the outer layers of cotton fiber.
Figure 1.2 illustrates schematically the distribution of cellulose and other non-cellulosic materials in the various layers of cotton fiber.. The outermost layer is the cuticle. It is a thin film of mostly fats and waxes. Owing to non-structured orientation of cellulose and non-cellulosic materials, the primary wall surfaces is unorganized and open. This gives the flexibility to the primary wall, which is required during the cell growth. The basic ingredients, that are responsible for complicated interconnections in the primary wall, are cellulose, hemicelluloses, pectins, proteins and ions. These components are present throughout the primary wall. The only difference is the concentration and nature of each component, as when approaching the secondary wall. In the secondary wall, only crystalline cellulose is present, which is highly ordered and has compact structure the cellulose fibrils in the secondary wall are laying parallel to one another.
Table 1.1 Typical composition of dry mature cotton fiber
| Constituents | Composition (%) | |
| Whole fiber | Outer layer | |
| Cellulose | 94 | 54 |
| Waxes | 0.6-1.3 | 14 |
| Pectic substances | 0.9-1.2 | 9 |
| Protein (nitrogen substances) | 0.6-1.3 | 8 |
| Ash | 1.2 | 3 |
| Organic Acid | 0.8 | – |
| Others | 1.4 | 12 |

1.5 The cuticle – the outermost layer
The aerial surfaces of vascular plants are covered with an extra cellular layer called the cuticle or cuticular membrane that overlays the cell wall of epidermal cells. The term “cotton waxes” has been used for all lipid compounds found in the cuticle of fiber. The main function ascribed to the cuticle is to minimize water losses from cotton fiber. Other functions are, to limit the loss of substances from fibers internal tissues, and to protect the fiber against physical, chemical and biological aggressions. The cuticle contains primary alcohols, higher fatty acids, hydrocarbons, aldehydes, glycerides, sterols, acyl components, resins, cutin and sobering, which are called waxes. The cuticle gives a soft touch to the fiber and reduces the friction forces during spinning. At the same time, presence of these waxy materials is Initiation of fiber growth detrimental in chemical processing of cotton yarn because it gives the fiber surface a high hydrophobicity. The cotton waxes are solid substances with rather high and wide range of melting points (64°C to 214°C) and constitute around 0.4 to 0.8% dry weight of raw cotton After treatment with boiling NaOH, waxes are hydrolyzed into a sodium salt of the fatty acid and alcohol. Apart from the above mentioned components in the cuticle, there are also some complex biopolymers present.
1.6 The primary wall
The primary wall in a cotton fiber is a thin film with a thickness of about 0.5 μm. It serves as the exterior surface of the fiber. In the primary wall, apart from amorphous cellulose, most of the constituents are non-cellulosic materials. This layer is flexible and swells uniformly in all directions. The typical composition of the primary wall with details of each component. the primary wall contains amorphous cellulose, pectins, proteins, hemicelluloses and coloring components. It is important to study the individual component and interconnections in the primary wall, for attacking specific components with enzymes to destabilize the primary wall efficiently. A brief description of the primary wall components, followed by a description of their interconnections is given below.
1.6.1 Details of each constituent in the primary wall
I. Cellulose:
Cellulose in the primary wall is heterogeneous and has a low degree of polymerization (DP up to 2000 glucose units) compared with cellulose in the secondary wall. The amorphous region of cellulose in a cotton fiber is characterized by its ability to swell in water. The orientation of cellulose macromolecules in the primary wall is low, that means that individual macromolecules are not arranged in any definite order. The cellulose micro fibrils in the primary wall are surrounded by a matrix of other non-celluloses. Micro fibrils of cellulose are crystalline aggregates of β(1-4)-linked glucose polymers. They are omnipresent elements of the plant cell wall and are responsible for much of its tensile strength.
II. Hemicelluloses:
Hemi cellulose is the name of a heterogeneous group of branched matrix forming polysaccharides. Hemicelluloses bind non-covalently to the surface of cellulose micro fibrils in the primary wall. They form a coating over the cellulose micro fibrils and are able to cross-link them into a complex network of the primary wall. There are several classes of hemicelluloses with an average 50 glucose units that are linearly β (1-4)-linked to one another. The difference between various classes of hemicelluloses is expressed in terms of oligosaccharide side chains
III. Glycoprotein:
Glycoprotein also known as extensions account up to 15% of the primary cell wall mass. Glycoprotein contains a protein backbone with extended rod like carbohydrates that protrude outwards. The carbohydrates in the glycoprotein account for 65% of the total structure. For a cotton fiber, these rod shape extensions are made up of roughly 300 amino acids, and abundantly contain hydroxyl-proline (Hyp). Most of the hydroxyl-prolines are glycosylated with chains of three or four sugar residues e.g. arabinose and galactose. The carboxyl-terminal peptides in the glycoprotein molecule are covalently linked by disulfide bonds and often contain an oligosaccharide chain. These oligosaccharides chains are also interconnected with other polysaccharides in the primary wall.
IV. Pectins:
Pectins are acidic polysaccharides, which are found in fruits, fibers and vegetables. Pectin being a non-cellulosic material in cotton fibers plays several important roles. It contributes to the firmness and structure of cotton fiber, both as a part of the primary cell wall. Pectin acts as cementing material for the cellulosic network in the primary wall. Pectin, as a hydrating agent, controls the movement of water and other plant fluids through the rapidly growing fiber.
V. Coloring matters:
Coloring matter (pigments) in cotton fibers is rarely studied. In the natural state, cotton is off-white, cream, brownish or greyish green, depending on the source and growing conditions. However these coloring components do not take part in the scouring process.
VI. Metal contents: The primary wall of a cotton fiber contains different quantities of metal depending on their growing conditions and source. Potassium is the most abundant metal ion in cotton fibers followed by magnesium and calcium. Other metal ions which are present in traces are sodium, iron, manganese, copper and zinc. Removal of calcium is essential during the scouring process for better primary wall destabilization. However, removal of rest of the metals from cotton fibers is also important, because they can contribute to problems during further wet-pretreatment processes like oxidative bleaching.
1.7 The secondary wall
Cellulose in the secondary wall is characterized by a higher degree of polymerization (nearly 5000 units) compared with cellulose in the primary wall. Table 2.4 shows the various units and sub-units of cotton cellulose with their diameter. In the secondary wall of a cotton fiber, two cellulose molecules can form a long planar chain of β(1-4)-linked glucose units, resulting in a ribbon like structure. These two cellulose chains forms a sheet, which is called an elementary fibril. Micro fibrils of cellulose are crystalline aggregates of approximately 21 elementary cells (Table 2.4). Intermolecular hydrogen bonds play an important role to stabilize chains of elementary fibril to become a micro fibril. Finally, a large numbers of such micro fibrils are laying in parallel direction forms the various layers of the secondary cell wall.

1.8 Wet-pretreatment process for the cotton textiles
Wet-pretreatment textile finishing forms the most important stage in the textile processing chain as shown in Figure 1.3. Wet textile processes are called ‘wet’ because they use water as the medium for transport of mass and heat across textile materials. Wet pre-treatment consists of desizing, scouring and bleaching. A short description of these three stages is given below.

Scouring – also known as boil off, kiering, kier boiling or caustization is the first process step in which the fiber surface is treated. The objective of a scouring process is to make the material hydrophilic, before it undergoes other processes like bleaching, dyeing and printing (Figure 1.4). A desired hydrophilicity during the scouring can be achieved by removing non-cellulosic material from the cotton, especially from the cuticle (waxes and fats) and the primary wall (e.g. pectin, protein and organic acids). More precisely scouring not only removes non-cellulosic material from cotton fibers but also removes substances that have adhered to the fibers during the production of the yarn or fabric. Substances like dirt, lint, pesticides, oils, and any sizing agent applied to yarns to facilitate weaving. Effective scouring is essential for subsequent processing of any cotton made substrate, regardless of its natural source.
1.9 Overview of the alkaline scouring for grey cotton
One of the earliest techniques for the scouring of cotton fabric involved the use of potash, the by-product of wood combustion. A major component of wood ash is potassium oxide, and when wood ash is added to water, the potassium oxide reacts to produce potassium hydroxide, a strong alkali. Early textile chemists prepared their fabric by treating the fabric in hot slurries of potash, followed by neutralization of the treated fabric in solutions of buttermilk. Although it can be argued that the chemicals used by ancient textile chemists were of ‘natural’ origin, the release of these natural substances did not have a positive impact on the environment. Soap is an example of a supporting scouring agent, before the introduction of synthetic detergents. The major drawback with soap as a supporting agent is its tendency to form insoluble calcium salts or ‘scum’ in hard water . Even today, alkaline scouring of cotton is still the most widespread commercial technique for removing or rupturing the fiber cuticle to make the fiber absorbent for the cotton processing. Various scouring agents used in the textile industry. Although sodium hydroxide is used generally for the scouring, sodium carbonate and calcium hydroxide are also mentioned as a scouring agent.
Scouring of cotton fabric is typically done with a hot solution (90°C to 100°C) of sodium hydroxide (± 1 mol/L) for up to one hour. The concentration of alkali used and the time and temperature conditions needed depend on the condition of the starting materials and the desired quality of the scoured fabric. Reducing agents are added during the scouring process to prevent oxidation of cellulose by air oxygen at high pH. Other chemicals for instance, wetting agents, emulsifying agents and chelating agents are also included in typical preparation baths for scouring. Wetting agents act by reducing the surface tension of water enabling improved penetration of the chemicals into the cotton fabric. Emulsifying agents assist in removing waxy materials. Chelating agents remove polyvalent metal ions such as calcium, magnesium, iron or other salts that can have a harmful effect on subsequent wet-processing operations. Polymeric materials can also act as chelating agents or as pickup enhancing agents for the application in continuous preparation processes. These various chemicals tend to be used in excessively high amounts. The key factors for a successful industrial scouring process are the concentration of the NaOH, the treatment temperature, the reaction time and the exclusion of air to avoid weakening the fiber by the formation of oxy-cellulose.
CHAPTER 2
SCOURING PROCEDURE
2.1 Scouring
Natural fibers contain oils, fats, waxes, minerals, leafy matter and motes as impurities that interfere with dyeing and finishing. Synthetic fibers contain producer spin finishes, coning oils and/or knitting oils. Mill grease used to lubricate processing equipment mill dirt, temporary fabric markings and the like may contaminate fabrics as they are being produced. The process of removing these impurities is called Scouring. Even though these impurities are not soluble in water, they can be removed by Extraction, dissolving the impurities in organic solvents, Emulsification, forming stable suspensions of the impurities in water and Saponification, Converting the contaminates into water soluble components.
2.2 Causes behind performing scouring process on cotton
- To make the fabric highly hydrophilic.
- To remove impurities such as fats and waxes, oils, pectin, protein etc. as completely as possible.
- To produce a clean material.
- To remove non-cellulosic substances of the cotton.
2.3 Materials
- Electronic Balance.
- Pipette.
- Measuring Cylinder
- IR Lab Dyeing Machine
- Oven Dryer
- Beaker
- Glass rod
2.4 Cotton Scouring
The soda-ash boil
- The type of alkali used for scouring of cotton depends on the quality of goods.
- For example, if colored yarns present in the fabric, sodium carbonate is ideally suited because of its low pH.
- Cotton yarns to be dyed in dark shade should be scoured with 1-2% sodium carbonate solution for 30 min in presence of wetting agent.
The mixture of caustic-soda and soda-ash boil
- 2 parts of caustic soda and 1 part of sodium carbonate
- Single stage boiling.
- Soda-ash softens the water while interacting with Ca and Mg salts (if such are present) .
- It creates an active reaction of the medium which is most favorable for the formation of stable emulsions and suspensions
- Increases fiber swelling,
- Thus contributing to the release of impurities from the fiber
- Neutralizes fatty acids contained in the fabric by soap formation;
- Reduces the adherence of detergents to wool in the alkaline medium conditions.
The soap/detergent- soda-ash boil
- This combination is comparably milder combination than that of caustic soda and hence ideally suitable for more delicate cloths and color woven goods compensating for the slower action of the milder alkali by the addition of a detergent.
- Anionic products like sodium alkyl sulphates and alkyl aromatic sulphonates and non-ionics like polyethoxylated compounds are used as detergent.
- Sometimes mixtures of anionic and non-ionic products are used.
- After scouring, washing is carried out by hot progressive rinsing while gradually decreasing the temperature in order to avoid break down of the emulsion and precipitation of the impurities onto the cotton.
- Washing is completed by treating the fabric in an acid solution to neutralize any alkali retained by the fabric.
2.5 Selection of Scouring Agent depends on
- Kind of fiber;
- Fabric type i.e. woven or knitted,
- Thickness of the fabric;
- Texturised or non-texturised
- And the extent of impurities present in the fiber.
- The selection of alkali is most important as free alkali can have a deleterious effect on certain fibers.
- In cotton alkali neutralizes the carboxyl group in cellulose and in pectin.
- The hydroxyl groups on the glucose units in cellulose are also weakly acidic.
2.6 Chemicals used in Scouring
- Caustic Soda
- Soda Ash
- Detergent
- Wetting agent
- Sequestering agent
2.6.1 Scouring with NaOH – mechanism of action
When caustic soda is used as the alkali, it is believed that the primary cell wall of the cotton fiber is broken to some extent, which also assists in the wetting process. When used at concentrations of about 0.7% to 1.5% w/v, at temperatures above the melting point of the cotton wax, some of the fatty acid part of the wax forms a soap with the caustic soda, the esters are saponified, and the products of the reactions emulsify the remaining constituents of the wax and help to suspend any impurities. Fats are derived from the trihydric alcohols. Waxes are derived from monohydric alcohols Here reaction between fat and alkali are as follows.

Caustic soda therefore plays a very important role in cotton preparation. Cotton fabrics arrive at mills with number of impurities. This includes motes, seed coat fragments, pesticides, dirt, chemical residues, metallic salts of various kinds, and immature fibers. The clear mechanism of action of alkaline scouring on various non-cellulosic materials in the cotton fiber is given in Table 1.2. Scouring with NaOH cleans by physical loosening fragments from the fabric. By dissolution of metallic salts and chemical residues. It softens and preconditions the seed coat fragment materials entrapped in the yarns and the fabrics. The dilute alkali swells the seed coat fragment material and opens up the cell structure to access the hydrogen peroxide in bleaching that takes place later. The swelling process helps to loosen the attachment of the seed coat fragments from the yarn and fibers. High temperature in the presence of sodium hydroxide melts some of the waxy material and converts some of it to a water-soluble form. It also converts non-cellulosic material (pectins, hemicelluloses and proteins) in the cuticle-primary wall to water soluble forms to effect removal.
More precisely, the scouring process is based on the reaction between cotton impurities and alkali hydroxide. Traditional scouring implies a certain alkali consumption that determines the minimum concentration of sodium hydroxide to be used. When sodium hydroxide is brought into contact with the cotton fabric, some of the alkali absorbs, since the hydroxyl groups of cellulose have a weak acidic character. So, at pH around 13-14, cellulose absorbs about 1% or 10 g/kg of sodium hydroxide. Alkali is also required to neutralize the carboxyl group of the pectins. About 0.5% of the sodium hydroxide concentration is required to change the pectins into water soluble salts of pectic or meta-pectic acid. Neutralization of the amino acids obtained by hydrolysis of the protein used around 1% of sodium hydroxide. It is evident that around 3% to 4% (±1 mol/L) of sodium hydroxide is necessary for the saponification of waxes and to maintain sufficient alkalinity. Literature reveals that fats are esters of fatty acids with glycerol and constitute around 37% to 47% of the total fat constituents. They have low melting points and are hydrolyzed into soaps and glycerol (saponification reaction) quite easily using an aqueous solution of NaOH. So, during the scouring process, the intra and intermolecular hydrogen bridges of the cellulose are cleaved and the polar hydroxyl groups of the polysaccharides are solvated. The fabric swells, and this facilitates transport of the impurities from the interior of the fiber to the outside.
Table: 2.1 The removal of impurities of cotton fiber during alkaline
| Impurities | Mechanism of impurities removal |
| Fats and waxes |
|
| Pectin and related substances |
|
| Proteins and amino acids |
|
| Hemicelluloses |
|
| Inorganic substances, minerals and heavy metals |
|
2.6.2 Soda ash
Sodium Carbonate (also known as washing soda, soda crystals or soda ash or “Soda Carbonate”), Na2CO3, is a sodium salt of carbonic acid. It most commonly effloresces to form a white powder, and is domestically well known for its everyday use as a water softener. It has a cooling alkaline taste, and can be extracted from the ashes of many plants. It is synthetically produced in large quantities from table salt in a process known as the Solvay process. Sodium carbonate is also used as a relatively strong base in various settings. For example, sodium carbonate is used as a pH regulator to maintain stable alkaline conditions necessary for the action of the majority of developing agents. It is a common additive in municipal pools used to neutralize the acidic effects of chlorine and raise pH.
In dyeing with fiber-reactive dyes, sodium carbonate (often under a name such as soda ash fixative or soda ash activator) is used to ensure proper chemical bonding of the dye with the fibers, typically before dyeing (for tie dyes), mixed with the dye (for dye painting), or after dyeing (for immersion dyeing).
2.6.3 Detergent
A detergent is a material used for cleaning. The term is sometimes used to differentiate between soap and other surfactants used for cleaning. The earliest detergent substance was undoubtedly water; after that, oils, abrasives such as wet sand, and wet clay. The oldest known detergent for washing. Other detergent surfactants came
- Surfactants to ‘cut’ (emulsify) grease and to wet surfaces.
- Abrasive to scour.
- Substances to modify pH or to affect performance or stability of other ingredients, acids for descaling or caustics to break down organic compounds.
- Water softeners to counteract the effect of “hardness” ions on other ingredients.
- Oxidants (oxidizers) for bleaching, disinfection, and breaking down organic compounds.
- Non-surfactant materials that keep dirt in suspension.
- Enzymes to digest proteins, fats, or carbohydrates in stains or to modify fabric feel.
- Ingredients that modify the foaming properties of the cleaning surfactants, to either stabilize or counteract foam.
- Ingredients to increase or decrease the viscosity of the solution, or to keep other ingredients in solution, in a detergent supplied as a water solution or gel
- Ingredients that affect aesthetic properties of the item to be cleaned, or of the detergent itself before or during use, such as optical brighteners, fabric softeners, colors, perfumes, etc.
- Ingredients such as corrosion inhibitors to counteract damage to equipment with which the detergent is used
- Ingredients to reduce harm or produce benefits to surface, when the detergent is used to clean.
- Preservatives to prevent spoilage of other ingredients.
2.6.4 Surfactant
Surfactants are wetting agents that lower the surface tension of a liquid, allowing easier spreading, and lowering of the interfacial tension between two liquids. The term surfactant is a blend of surface active agent. Surfactants are usually organic compounds that are amphiphilic, meaning they contain both hydrophobic groups (their tails) and hydrophilic groups (their heads). Therefore, they are soluble in both organic solvents and water.
Properties
Surfactants reduce the surface tension of water by adsorbing at the liquid-gas interface. They also reduce the interfacial tension between oil and water by adsorbing at the liquid-liquid interface. Many surfactants can also assemble in the bulk solution into aggregates. Examples of such aggregates are vesicles and micelles. The concentration at which surfactants begin to form micelles is known as the critical micelle concentration (CMC). When micelles form in water, their tails form a core that can encapsulate an oil droplet, and their (ionic/polar) heads form an outer shell that maintains favorable contact with water. When surfactants assemble in oil, the aggregate is referred to as a reverse micelle. In a reverse micelle, the heads are in the core and the tails maintain favorable contact with oil. Surfactants are also often classified into four primary groups; anionic, cationic, non-ionic, and zwitterionic (dual charge).
Thermodynamics of the surfactant systems are of great importance, theoretically and practically. This is because surfactant systems represent systems between ordered and disordered states of matter. Surfactant solutions may contain an ordered phase (micelles) and a disordered phase (free surfactant molecules and/or ions in the solution). Ordinary washing up (dishwashing) detergent, for example, will promote water penetration in soil, but the effect would only last a few days (many standard laundry detergent powders contain levels of chemicals such as alkali and chelating agents, which can be damaging to plants and should not be applied to soils). Commercial soil wetting agents will continue to work for a considerable period, but they will eventually be degraded by soil micro-organisms. Some can, however, interfere with the life-cycles of some aquatic organisms, so care should be taken to prevent run-off of these products into streams, and excess product should not be washed down.
2.6.5 Sequestering Agents/ Chelating agent
Chelation is the formation or presence of two or more separate bindings between a polydentate (multiple bonded) ligand and a single central atom. Usually these ligands are organic compounds, and are called chelants, chelators, chelating agents, or sequestering agents. The ligand forms a chelate complex with the substrate. Chelate complexes are contrasted with coordination complexes with monodentate ligands, which form only one bond with the central atom. Chelants, according to ASTM-A-380, are “chemicals that form soluble, complex molecules with certain metal ions, inactivating the ions so that they cannot normally react with other elements or ions to produce precipitates or scale.”
The functions of the these agents in the soap and detergent formulations are for the prevention of –
- Film and scum formation,
- Precipitation of hard water,
- Calcium and magnesium inhibition of foaming properties,
- Clogging of liquid dispersions.
2.7 Process descriptio
Recipe
- Caustic Soda (NaOH): X g/l
- Soda Ash: 2g/l
- Sequestering Agent: 1g/l
- Wetting Agent: 1g/l
- Detergent: 1g/l
- Material Liquor Ratio: 1:20
- Temperature: 105oC
- Time: 40min

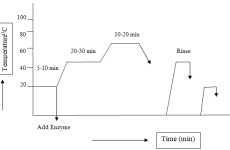
All the chemicals were taken according to recipe measuring by electronic balance. Stock solution of caustic soda of 2% & 4% and soda ash of 1% were prepared. Before putting the samples in scouring bath, the samples were kept in woven dryer at 1050C for 30minutes to measure woven dry weight. After taking woven dry weight of each sample, those were put in a water bath. Then the chemicals and water were taken according to recipe and later on the samples were given in scouring bath. Completing all this, each pot was given in the machine. The program was set for 40min at 105oC. Unloading the samples hot wash was done with detergent for 10min at 80oC. Finally cold wash was done and again the samples were dried in woven dryer at 105oC for 30min and estimating the scouring effect of cotton were measured.
CHAPTER 3
SCOURING PARAMETERS
3.1 Introduction
The scouring process depends on many factors such as the type, color and cleanliness of the cotton material, the twist and count of the yarn, the construction of the fabric etc. The impurities which are most difficult to remove are wax, seed fragments and oil strains. Scouring may be carried out on loose fibers, slivers, yarns or fabric. But for ease of handling fabric is preferred.
3.2 Raw Materials and other inputs
1. Water Quality
- Hardness
- Bi-carbonate
- Carbonates sulphates and chloride
- TDS (Total Dissolved Solids)
- Turbidity
- Other metals –iron, copper
2. Quality of Auxiliary Chemicals used.
b. Quality of surfactants like wetting, scouring agents
- Wetting time of wetting agents
- Solid content
- Ionic nature
- Cloud point of non-ionic products
- Foaming characters
c. Quality of sequestering agents
- Chelating power
- pH
- Solid content
3.3. Quality of Basic Chemicals used and its concentration
- Caustic soda – purity and concentration percentage
- Soda Ash – purity and concentration
3.4 Effect of Time and Temperature
Time and temperature of scouring are interrelated. As the temperature increases, less time is needed. The scouring performance is accelerating at high temperature.
3.5 Concentration
Concentration is also interrelated with time and temperature. Higher concentrations require less time and temperature.
3.6 Effect of PH
PH has a profound effect on scouring with caustic soda. The optimum PH for scouring is between 10.5 and 11.5.
3.7 Material liquor ratio
Higher material liquor needs high time and temperature. It also needs more chemical, later on more load on ETP. Overall cost also become high due to high material liquor ratio and vice versa.
CHAPTER 4
RESULT & DISCUSSION
4.1 Introduction
The main changes which occur in cotton goods during scouring process are loss in weight (4-8%), loss in length due to shrinkage during boiling treatment, alteration in yarn count affected by both losses and changes in strength. However, the most important characteristic of scoured fabric is increased wet ability, which is necessary for subsequent processing. Wettability must be obtained not only in the capillary spaces between fibers, but also inside the fiber themselves. While scouring is beneficial for wettability, when carried out in severe conditions, it includes fiber deterioration e.g. the creation of cavities in fibers or dissolution of the cuticle and primary wall.
4.2 Estimation of scouring effect on cotton
The scouring effect of cotton can be estimated by carrying out by the following tests:
1. Determination of weight loss.
2. Absorbency test-
- Immersion test.
- Drop test and
- Spot test.
4.2.1 Determination of weight loss
The loss in weight of fabric during scouring shows that a considerable amount of impurities are removed. The weight of unscoured and scoured samples taken separately at the same moisture content and then the weight loss is measured in percentage.
……………………….Weight (before scouring – after scouring)
Weight loss=………………………………………………………………………………….. x 100%
…………………………………….Weight before scouring
4.2.1.1 Weight loss of Single Jersey (Plain) Fabric
Table: 4.1 Weight Loss (%) of single jersey (plain) fabric
| Sample No | Caustic soda(g/l) | Before Scoured Weight(gm) | After Scoured Weight(gm) | Weight Loss (%) |
| 01 | 1 | 5.05 | 4.88 | 3.37 |
| 02 | 2 | 5.03 | 4.82 | 4.17 |
| 03 | 3 | 5.00 | 4.77 | 4.60 |
| 04 | 4 | 4.95 | 4.70 | 5.05 |
| 05 | 5 | 5.08 | 4.82 | 5.20 |
| 06 | 6 | 5.00 | 4.73 | 5.39 |
| 07 | 7 | 4.97 | 4.72 | 5.30 |
| 08 | 8 | 5.00 | 4.70 | 6.00 |
| 09 | 9 | 5.00 | 4.67 | 6.60 |
| 10 | 10 | 5.00 | 4.64 | 7.20 |
| 11 | 11 | 5.00 | 4.63 | 7.40 |
| 12 | 12 | 4.93 | 4.57 | 7.30 |

4.2.1.2 Weight loss of Single Jersey (Fleece) Fabric
Table: 4.2 Weight Loss (%) of Single Jersey (Fleece) fabric.
| Sample No | Caustic soda(g/l) | Before Scoured Weight(gm) | After Scoured Weight(gm) | Weight Loss (%) |
| 01 | 1 | 5.19 | 5.00 | 3.67 |
| 02 | 2 | 5.01 | 4.80 | 4.19 |
| 03 | 3 | 5.03 | 4.79 | 4.77 |
| 04 | 4 | 5.08 | 4.82 | 5.12 |
| 05 | 5 | 4.95 | 4.68 | 5.45 |
| 06 | 6 | 4.97 | 4.68 | 5.83 |
| 07 | 7 | 5.01 | 4.70 | 6.17 |
| 08 | 8 | 5.12 | 4.81 | 6.05 |
| 09 | 9 | 5.04 | 4.72 | 6.35 |
| 10 | 10 | 4.93 | 4.58 | 7.10 |
| 11 | 11 | 4.97 | 4.61 | 7.24 |
| 12 | 12 | 5.05 | 4.67 | 7.50 |

4.2.1.3 Weight Loss (%) of 1×1 Rib fabric
Table: 4.3 Weight Loss(%) of double jersey (1×1 Rib) fabric
| Sample No | Caustic soda (g/l) | Before Scoured Weight (gm) | After Scoured Weight (gm) | Weight Loss (%) |
| 01 | 1 | 5.08 | 4.90 | 3.54 |
| 02 | 2 | 5.02 | 4.81 | 4.18 |
| 03 | 3 | 5.09 | 4.86 | 4.52 |
| 04 | 4 | 5.06 | 4.82 | 4.71 |
| 05 | 5 | 4.95 | 4.70 | 5.03 |
| 06 | 6 | 4.92 | 4.65 | 5.49 |
| 07 | 7 | 4.98 | 4.68 | 6.02 |
| 08 | 8 | 5.01 | 4.69 | 6.39 |
| 09 | 9 | 5.12 | 4.80 | 6.25 |
| 10 | 10 | 5.03 | 4.68 | 6.96 |
| 11 | 11 | 5.01 | 4.67 | 6.78 |
| 12 | 12 | 5.07 | 4.71 | 7.05 |

4.2.1.4 Comparison of weight loss (%) among single jersey (plain), single jersey (fleece) & double jersey (1×1 Rib) versus Caustic soda

4.2.2 Absorbency
a. Immersion test-
Test:- Sample of 1cm x 1cm size is cut and it is left on water surface. With the help of stop watch, the time of fabric for immersing is recorded.
Result:- The standard time of immersing is 5sec.
b. Drop test:
In a pipette a solution of 1% direct red or congo red is taken and droplet of solution put on the places of the fabric such that not spread. Then the absorption of the colored drop is observed visually.
The standard time for the absorption of one drop of solution is 0.- 0.8sec.
c. Spot test;
In a pipette a solution of 1% direct red or congo red is taken and droplet of solution put on the different places of the fabric. Then the shape of the absorbed area on the fabric is observed.
4.2.2.1 For Single Jersey Fabric (Plain of GSM-170)
Table: 4.4 Effect of Scouring on Single Jersey (plain) Fabric
| Sample No | Caustic Soda (g/l) | Effect of Scouring | ||
| Drop Test | Immersion Time | Spot Test | ||
| 01 | 1 | 0.7 sec | 0.5sec | |
| 02 | 2 | 0.6 sec | 0.5sec | |
| 03 | 3 | 0.5 sec | 0.5sec |  |
| 04 | 4 | 0.5 sec | 0.5sec | |
| 05 | 5 | 0.5 sec | 0.5sec | |
| 06 | 6 | 0.5 sec | 0.5sec | |
| 07 | 7 | 0.5 sec | 0.5sec | |
| 08 | 8 | 0.5 sec | 0.5sec | |
| 09 | 9 | 0.5 sec | 0.5sec | |
| 10 | 10 | 0.5 sec | 0.5sec | |
| 11 | 11 | 0.5 sec | 0.5sec | |
| 12 | 12 | 0.5 sec | 0.5sec | |
4.2.2.2 For Single Jersey Fabric (fleece GSM-310)
Table: 4.5 Effect of Scouring on Single Jersey Fabric (Fleece)
| Sample No | Caustic Soda (g/l) | Effect of Scouring | ||
| Drop test | Immersion Time | Spot Test | ||
| 01 | 1 | 0.7 sec | 0.6sec | |
| 02 | 2 | 0.6 sec | 0.5sec | |
| 03 | 3 | 0.5 sec | 0.5sec | |
| 04 | 4 | 0.5 sec | 0.5sec | |
| 05 | 5 | 0.5 sec | 0.5sec | |
| 06 | 6 | 0.5 sec | 0.5sec | |
| 07 | 7 | 0.5 sec | 0.5sec | |
| 08 | 8 | 0.5 sec | 0.5sec | |
| 09 | 9 | 0.5 sec | 0.5sec | |
| 10 | 10 | 0.5 sec | 0.5sec | |
| 11 | 11 | 0.5 sec | 0.5sec | |
| 12 | 12 | 0.5 sec | 0.5sec | |
4.2.2.3 Double Jersey (1×1 Rib) Fabric
Table: 4.6 Effect of Scouring on double jersey (1×1 Rib) Fabric.
| Sample No | Caustic Soda (g/l) | Effect of Scouring | ||
| Drop test | Immersion Time | Spots Test | ||
| 01 | 1 | 0.7 sec | 0.6sec | |
| 02 | 2 | 0.6 sec | 0.5sec | |
| 03 | 3 | 0.5 sec | 0.5sec | |
| 04 | 4 | 0.5 sec | 0.5sec | |
| 05 | 5 | 0.5 sec | 0.5sec | |
| 06 | 6 | 0.5 sec | 0.5sec | |
| 07 | 7 | 0.5 sec | 0.5sec | |
| 08 | 8 | 0.5 sec | 0.5sec | |
| 09 | 9 | 0.5 sec | 0.5sec | |
| 10 | 10 | 0.5 sec | 0.5sec | |
| 11 | 11 | 0.5 sec | 0.5sec | |
| 12 | 12 | 0.5 sec | 0.5sec | |
4.3 Findings
- The weight loss(%) is increasing with respect to increasing of caustic soda upto a certain limit but later on if the caustic soda is increased the weight loss(%) is not so high.
- Minimum weight loss and maximum absorbency can be obtained up to 2-4 g/l of caustic soda for all fabric.
- Without wetting agent absorbency is not obtained prefect. As chemicals can not penetrate to interior to the fabric and waxes are not removed well.
- Fabric roughness will be increased by increasing higher amount of caustic soda in scouring bath.
- Final comment of standard recipe for different fabric with minimum caustic, minimum weight loss but maximum scouring effect on cotton is
- Caustic soda: 2—4 g/l for all type of fabric.
- Soda ash: 1—2.5 g/l.
- Wetting agent: 0.5—1 g/l
- Sequestering agent: 0.5—1 g/l
- Anticreasing agent: 0.5—1 g/l
- Time: 40 minutes
- Temperature: 1050c
- pH: 10.5—11.5
4.4 Drawbacks associated with the alkaline scouring
The scouring process requires large quantities of chemicals, energy and water and is rather time consuming. Owing to the high sodium hydroxide concentration and its corrosive nature, intensive rinsing is required that leads to a high water consumption. The use of high concentrations of sodium hydroxide also requires the neutralization of wastewater, which requires additional acid chemicals. Furthermore, the alkaline effluent requires special handling because of very high BOD and COD values. Apart from the above wet processing problems, the biggest drawback of alkaline scouring is a non-specific degradation of cellulose that produces fabrics of lower tensile strength and therefore of lower quality. Moreover alkaline scouring is hazardous to the workers and creates an unpleasant work atmosphere. Although, alkaline scouring is effective and the costs of NaOH are low, this process can be improved considerably to meet today’s energy and environmental demands. Alkaline scoured fabric becomes harsh using more NaOH.
CHAPTER 5
CONCLUSION
5.1 Conclusions
The major goal of any scouring process is to improve the water absorbency of natural fibers by removing the water-repellent components of the fiber cuticle which facilitates uniform dyeing and finishing. Since the cuticle is cross-linked to the primary cell wall by esterified pectic substances, efficient scouring correlates with a considerable removal of both waxy and pectic substances. Absorbency is quantitatively estimated by measuring the time required for a water drop to be absorbed or the distance traveled by a waterfront after a predetermined time. Although simple to perform, these techniques lack accuracy and cannot discern between highly efficient scouring techniques, especially when the fiber becomes rapidly wettable and insignificant times are being recorded. At the opposite extreme are staining techniques targeted to bind to the newly accessible hydrophilic components, such as congo red for cellulose, the exposed cellulose correlated with an increase in the fabric’s hydrophilicity, from what a fabric’s weight loss was observed as function of the severity of the scouring treatment applied, clearly indicating that the bound caustic increases proportionally with the amount of available binding sites.
Scouring is related to hydrophilicity and can be achieved by uncovering the pores that are already present in the fibers, by removing waxes and other non-cellulosic materials in the primary wall. The technical feasibility of caustic scouring has been recognised by many researchers over the last decade. However, continuous Caustic scouring process has been widely implemented by textile industries. The most important reason identified was the ability to remove cotton fiber waxes during caustic souring. The pre-rinsing in hot water above 90°C with a surfactant helps to reduce the wax impurity load and renders a better subsequent effect towards primary wall destabilization. With these results we have recommended the usual scouring temperature 105°C for NaOH.
5.2 Outlook
Clear guidelines for the successful scouring process, which has proved on the laboratory scale. These guidelines are:
- Wax removal is a pre-requisite, since scouring is related to hydrophilicity and not how much pectin is removed.
- Efficient wax removal facilitates fast pectinase incubation and improves pectin removal rate and hence fast primary wall destabilisation, and
- Efficient wetting and mass transfer improvement are essential to convert slow diffusion based scouring process into faster process. In order to develop an industrial scouring process for cotton fabric based upon the guidelines we have given, more research is needed to be done in the future.
References
- Choudhury, Asim Kumar Roy, “Textile Preparation and dyeing” ISBN 81-204-1701-1 Delhi, India. Oxford & IBH Publishing(2006)
- Tomasino, Dr. Charles, “Chemistry & Technology of Fabric Preparation & Finishing” North Carolina, USA.
- Trotman, E. R, “Dyeing and Chemical Technology of Textile Fibers”, Fifth edition, London, UK, Charles Griffin
- Ghol, E.P.G. and Vilensky L.D. “ Textile Science”. Delhi, India 2nd edition, CBS Publishers (1983).
- Karmakar, S.R., “Chemical technology in the pretreatment processes of textiles”,Textile Science and Technology Series, 1st Ed., ISBN 0-444-500060-X, Elesevier Science B.V., Amsterdam, the Netherlands, 12, (1999)
- Rouette, H.K., Encyclopedia of Textile Finishing, Springer-Berlin, 1-3, ISBN 3-540-65031-8, Springer-Berlin, Germany, (2001).
- Walton, T.J., “Waxes, cutin and suberin”, Methods in Plant Biochemistry, 4, 105-158 (1990).
- Kissa, E., “Detergency: theory and technology”, Surfactant science series, Eds.W. Gale Cutler, M. Dekker, New York, USA, ISBN 0-8247-7503-1, 20, (1987
- Rosen, M.J., “Surfactant and Interfacial phenomena”, 2nd Ed. John Willey and Sons, NY, USA, (1978).
Founder & Editor of Textile Learner. He is a Textile Consultant, Blogger & Entrepreneur. Mr. Kiron is working as a textile consultant in several local and international companies. He is also a contributor of Wikipedia.