Treatment of Cotton Fabric with Chitosan for the Improvement of Exhaustion during Dyeing with Reactive Dye
Mustaque Ahammed Mamun1 &
Md. Ismail Hossain
Department of Textile Engineering
Dhaka University of Engineering & Technology (DUET)
Email: [email protected]1
ABSTRACT
This study presents the possibility of improvement of dye exhaustion during dyeing of cotton fabric with reactive dyes after treatment with chitosan. This experiment presents a new way to dye the cotton fabric with reactive dye after treating with chitosan. Cotton fabric was treated with chitosan solution at different concentrations (0.5%, 1%, 1.5%, 2.5% and 3%) and then dyed with reactive dye. The depth and fastness of shade of dyed fabric were analyzed by comparing the chitosan treated samples with untreated dyed fabric samples. It has been found that, the dyebath exhaustion is increased with the increment of chitosan concentrations. The exhaustion percentages have found 77.72%, 81.48%, 84.24%, 85.87% and 88.21% for the fabric treated with 0.5%, 1%, 1.5%, 2.5% and 3% chitosan solution respectively, while the exhaustion of dyebath is only 75.54% for untreated fabric. The K/S values (at λmax = 540 nm) of dyed samples have found 4.01, 4.25, 4.6, 4.8, 5.2 and 3.8 for 0.5%, 1%, 1.5%, 2.5%, 3% and untreated fabric respectively. The color fastness to washing and rubbing of the dyed fabrics was also evaluated. In case of dry rubbing, both types of fabric have shown almost similar fastness ratings. However, chitosan treated fabrics have shown inferior fastness rating in case of wet rubbing, particularly for the fabrics at higher chitosan concentrations.
Keywords: Chitosan, dyeing, cotton fiber, reactive dye
1. INTRODUCTION
1.1 Background
Cotton fiber is the most useable fiber (E.R. trotman et al. 1975). About 90 countries in the world used cotton fiber for various purposes. This fiber is used due to some reason such as one of the first advantages of cotton is comfort. Cotton is softer so it will it feel much more comfortable to wear. The advantages of cotton include inexpensive, there is an unlimited supply. It can be worn in the summer (warm days), or winter (cool days). Cotton also absorbs water and moisture easily. The uses of it are wearing apparel, home furnishing, hygiene uses, and eventually medical uses. It is a natural fiber so it is hypo allergenic, biodegradable, easily cleaned, breathable, light and easily colored. It can be dyed by using various colors or dyestuffs (M. Lewin et al., 2010).
Cotton fiber can be dyed with various dyes such as direct dye, vat dye, sulphur dye, mordant dye, azoic dye, reactive dye. The wash fastness of direct dye is not good (2-3) (E.R. trotman et al., 1975). Rubbing fastness of vat dye is not also good. Dyeing procedure of vat dye is difficult. It is costly. Sulphur dye is unhygienic for environment. Shading is not more than 10%. Limited range of color. Bronziness and Tendering effect are common defects in sulphur dyeing. The azoic dyeing procedure is complicated and time-consuming application procedures. On the other hand, reactive dyes have good fastness properties owing to the covalent bonding that occurs during dyeing (Broadbent et al. 2001).
Reactive dyeing is now the most important method for the coloration of cellulosic fibres. Cotton is made of cellulose molecules which react with the dye. During reactive dyeing the hydrogen atom in the cellulose molecule combines with the chlorine (Cl2) atom in the dyeing process and results in a bond. Reactive dyes used to dye cellulosic fibres. The dyes contain a reactive group, either a haloheterocycle or an activated double bond, that, when applied to a fibre in an alkaline dye bath, forms a chemical covalent bond with a hydroxyl group on the cellulosic fibre. There are some benefits, because the reactive dye consists with chromophores of dye part, bridging groups, reactive group bearing parts and reactive groups. Reactive groups are the responsible for the fixation on alkali condition. All of the reactive dyes are organic compounds. Organic compounds are much more harmful than the others.
The main disadvantage of reactive dyes is hydrolysis of reactive dye during the application of reactive dyes to cellulose fibers under highly alkaline conditions, a competing hydrolysis reaction takes place, originating in the non-reactive oxi-dye form, which is lost for dyeing and passes into the waste water. Unfixed or hydrolyzed reactive dye has to be washed off thoroughly in order to achieve the desired superior wet fastness of the reactive dyeing. As much as 50% of the total cost 0f a reactive dyeing process must be attributed to the washing off stages treatment of the resulting effluent. This aspect of the process should be recognized a major limitation that prevents reactive dyes from achieving the degree of success that was predicted for them at the time of their discovery. This hydrolysed dye is discharged as colored effluent effluent cost is risen up. Reactive dye hydrolysates are not easily adsorbed by sewage sludge in abiological clarification plant (D Fiebig et al., 1998). Moreover color is not easily removed by effluent treatment processes and in many cases the dyes are not readily biodegradable.this unhydrolysed dyes may pose an environment hazard. So, for decreasing hydrolysis salt is used but recently a compound is used before dyeing for increasing absorption which name is chitosan.
Chitosan is a non-toxic and chemically reactive, and biocompatible natural functional polymer, and has long been used as a biopolymer and natural material in many fields. Recently, chitosan effects widely studied are antistatic, bacteriostatic, biocompatibility properties conferred on various textiles (M. Sundrarajan et al. 2010). According to some previous experiences, wrinkle-resistance finishing with chitosan for fabric can improve its wrinkle degree but its handle and mechanical property got worse to a certain extent. Chitosan adsorption also increased the moisture absorption of the fibers. Chitosan with lower molecular weight increased the hydrophilicity of the treated fibers, but chitosan with higher molecular weight, decreased it.
1.2 Objectives of the thesis
The main objectives of the study to increase the dyeability reactivity dye by using eco-friendly biodegradable chitosan.
- To get desired exhaustion of dye in dyeing of cotton fabric with reactive dyes by using salt.
- To improve depth of shade cotton fabric by using chitosan.
- To improve the dye uptake and also to reduce the effluent load.
- To decrease the rate of hydrolysis of reactive dye.
- To improve the crease recovery of cotton fabric.
2. LITERATURE REVIEW
2.1 Cotton Fiber
Cotton is unique in nature which has the highest percentage of cellulose. It varies from 90% to 95%. This concentration depends upon many factors, such that maturity level and variety of cotton, place on cottonseed from where it is drawn. In cotton fiber there is certain non-cellulosic matter, which plays important role in growth and on forth coming processes. These are located either on the surface of the fiber or in center of fiber means inside the lumen. Being a natural fiber, there are many factors which affect the structure of cotton cellulose. There are primary and secondary walls in the cotton cellulose. Primary wall or outer wall is less crystalline and has less percentage of cellulose. Whereas, secondary wall is around the lumen and consist of pure cellulose cotton fiber is composed of the crystalline and amorphous structure.
2.1.1 Chemical Structure of Cotton Fiber
The molecular structure of cellulose has always been of great interest to scientists and over time several structures have been proposed. The linear polymer, β-D-glucopyronose with 1,4-glycosidic bonds, is the widely accepted structure for cellulose. Consequently, it may be considered as a polyhydric alcohol. Each glucopyranose ring in the cellulose chain contains three hydroxyl groups, a primary hydroxyl group in the 6- position and secondary hydroxyl groups in the 2- and 3- positions.

2.1.2 Cotton Fiber Ingredients
The basic ingredients, that are responsible for complicated interconnections in the primary wall, are cellulose, hemicelluloses, pectins, proteins and ions. These components are present throughout the primary wall. The only difference is the concentration and nature of each component, as when approaching the secondary wall. In the secondary wall, only crystalline cellulose is present, which is highly ordered and has compact structure the cellulose fibrils in the secondary wall are laying parallel to one another.
Table 2.1: Typical Composition of Dry Mature Cotton Fiber
| Constituents | Composition (%) | |
| Whole fiber | Outer layer | |
| Cellulose | 94 | 54 |
| Waxes | 0.6-1.3 | 14 |
| Pectin substances | 0.9-1.2 | 9 |
| Protein (nitrogensubstances) | 0.6-1.3 | 8 |
| Ash | 1.2 | 3 |
| Organic Acid | 0.8 | – |
| Others | 1.4 12 | |
2.1.3 Chemistry of Cotton Cellulose
Cellulose is an insoluble substance and mainly composed of polysaccharide, which holds chains of glucose monomers. It is only soluble in some specific solvents. It is the main constituent of plant cell walls and as well as of vegetable fibers. Cotton is one of the vegetable fibers which have the highest percentage of cellulose. There is a diverse structures and compositions of cellulose of cotton. Its structure makes it divergent from other naturally occurring matters. Cotton cellulose is unique in many ways and possesses distinct characteristics which make it highly useful for many purposes. It has been assumed that cotton cellulose structure is based on glucose unit only. However, it is also believed that there is modest amount of pentose is present, which is removed during scouring process. There is no evidence of the presence of pentose. Cotton cellulose is highly crystalline in nature and well oriented and has along and rigid molecular structure. The 1,4-D glucopyranose are the principle building blocks of cotton cellulose chain and are linked by l,4-glucodic bonds. Free rotation of the anhydrogluco-pyranose C-O-C link is stopped by steric effects. There are three hydroxyl groups attached to each anhydroglucose. One group is attached at C-6 and two at C-2 and C-3. Due to the presence of hydroxyl groups and the chain conformation, there are many more bonds possible (inter molecule and intramolecular). Such bonds make the fiber more rigid by increasing the rigidity of the structure of cotton cellulose.
2.1.4 Cross Section of Cotton Fiber
The cell wall is a dynamic structure which composition and form can change markedly, not only during cell growth but also after the cells have become matured. The cotton fiber is structurally built up into concentric zones and a hollow central core known as the lumen.

The mature fiber essentially consists of (from outside to inside) – the cuticle i.e. the outermost layer, the primary cell wall, the secondary wall and the lumen. Figure 1.1 systematically shows the different layers present in the cotton fiber with the compositions of each layer. Cotton contains nearly 90% of cellulose and around 10% of non-cellulosic substances, which are mainly located in the cuticle and primary wall of the fiber. Typical components in dry mature cotton fibers most of the non-cellulosic materials are present in the outer layers of cotton fiber.
Figure illustrates schematically the distribution of cellulose and other non-cellulosic materials in the various layers of cotton fiber. The outermost layer is the cuticle. It is a thin film of mostly fats and waxes. Owing to non-structured orientation of cellulose and non-cellulosic materials, the primary wall surfaces is unorganized and open. This gives the flexibility to the primary wall, which is required during the cell growth.
2.2 Reactive Dye
A dye, which is capable of reacting chemically with a substrate to form a covalent dye substrate linkage, is known as reactive dye. Here the dye contains a reactive group and this reactive group makes covalent bond with the fibre polymer and act as an integral part of fibre. This covalent bond is formed between the dye molecules and the terminal –OH (hydroxyl) group of cellulosic fibres on between the dye molecules and the terminal –NH2 (amino) group of polyamides or wool fibres. Reactive dyes in the simplest terms, all reactive dyes are made up of three basic units, a chromophore, a bridge and a reactive group/ groups (either a haloheterocycle or an activated double bond). One problem is that instead of reacting with the -OH groups on the cellulose, the fibre-reactive group may react with the HO- ions in the alkali solution and become hydrolysed. The hydrolysed dye cannot react further. This must be washed out of the fabric before use.
2.2.1 Properties of Reactive Dye:
- Reactive dyes are anionic dyes, which are used for dyeing cellulose, protein and polyamide fibres.
- Reactive dyes are found in power, liquid and print paste form.
- During dyeing the reactive group of this dye forms covalent bond with fibre polymer and becomes an integral part of the fibre.
- Reactive dyes are soluble in water.
- They have very good light fastness with rating about 6. The dyes have very stable electron arrangement and can protect the degrading effect of ultra-violet ray.
- Textile materials dyed with reactive dyes have very good wash fastness with rating Reactive dye gives brighter shades and have moderate rubbing fastness.
- Dyeing method of reactive dyes is easy. It requires less time and low temperature for dyeing.
- Reactive dyes are comparatively cheap.
- Reactive dyes have good perspiration fastness with rating 4-5.
- Reactive dyes have good perspiration fastness.
2.2.2 The Components of Reactive Dye

2.2.3 Chemical Reactions between Reactive Dyes and Fibers:
a) Nucleophilic Substitution:
Nucleophilic substitution characterizes dye-fiber fixation that occurs when a leaving group in the reactive system is displaced as a result of an interaction with a nucleophilic group on the polymer chain. Nucleophilic substitution is facilitated by the electron withdrawing properties of the aromatic nitrogens, and the chlorine, and the anionic intermediate is resonance stabilised as well. This resonance means that the negative charge is delocalised onto the electronegative nitrogens.
For example:

But this requires harsh conditions. To improve the rate under mild conditions, spowerful electron-withdrawing groups such as -NO2 may be added.

However, this will only work if there is a good leaving group, such as -Cl or -N2.
The major fibre-reactive group which reacts this way contains six-membered, heterocyclic, aromatic rings, with halogen substituents.
For example, the Procion dye-

Where X = Cl, NHR, OR.
The reaction of a monochlorotriazine reactive dye with a hydroxy group of cellulose is typical of this process.
a) b)
b)

The same process accounts for the competitive hydrolysis reaction between the dyes and water during dye application.
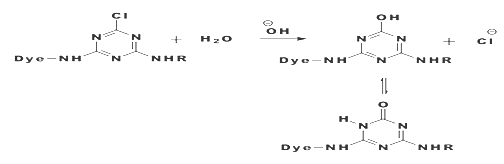
b) Nucleophilic Addition:
Nucleophilic addition characterizes the dye-fiber reaction in which a nucleophilic group in the fiber adds across an activated carbon-carbon double bond in the reactive group. Most of reactive systems used contain a vinylsulphone moiety. The vinylsulphone reactive group itself is usually not present in commercial form of the dyes employed. Instead, more stable precursor such as the β-sulphatoethylsulphone group is used. The two-stagenprocess associated with fiber fixation is structurally related dyes containing a β-sulphatoethylsulphamoyl group probably form a cyclic compound capable of reacting with cellulose to give cellulose ether.
Systems based on activated double bonds also undergo a competitive hydrolysis reaction.


2.3 Chitin
Chitin is the second most abundant polysaccharide on earth after cellulose. It is found in the shells of crustaceans such as crabs, shrimps and lobster, in the exoskeleton of insectsand molluscs and in the cell walls of certain fungi. Chitin was first isolated by Braconnot in 1811 and called it fungine. Later, in 1823, Odier found it in insects and named it as chitin. Chitin is mostly produced from crab and shrimp shells which are dumped as huge wastes by the seafood industry. The crustacean shell waste contains 30-50% calcium carbonate, 30-40% protein and 20-30% chitin on a dry mass basis.
2.3.1 Structure of Chitin, Chitosan and Cellulose
Chemically, Chitin is a homopolymer of 2-acetamido-2-deoxy-β-D-glucopyranose with some of the glucopyranose residues existing as 2-amino-2-deoxy-β-Dglucopyranose. Its molecular weight, purity, and crystal morphology are dependent on their sources. It occurs naturally as one of three crystalline polymorphic forms; α-, β- or γ-chitin differing in chain packing in crystalline regions. α-chitin has anti-parallel chains, β-chitin has a parallel stack structure while arrangement of two parallel chains and one anti-parallel chain has been suggested for γ-chitin. Both α- and β-chitins possess C=O·····H−N intermolecular hydrogen bonds, but the intermolecular hydrogen bonds between −CH2OH groups are present in the α-chitin and absent in the β-chitin. Therefore, β-chitin swells easily in water to produce hydrates unlike α-chitin, which has a strong three-dimensional hydrogen bond network. β-chitin is found in squid and marine diatoms and is rare while α-chitin is most abundantly found in crustaceans, insects, and fungi. Hence, chitosan is commercially prepared from α-chitin. The molecular structure of chitin, chitosan and cellulose are shown in Figure 2.8.

Chitin, chitosan and cellulose are naturally occurring β-1, 4-linked linear polysaccharides. Chitin has the same backbone as cellulose, but it has an acetamide group on the C-2 position instead of a hydroxyl group. Chitin is a homopolymer of 2-acetamido-2-deoxy-β-D-glucopyranose, although some of the glucopyranose residues exist as 2-amino-2-deoxy-β-Dglucopyranose. Chitosan is the N-deacetylated derivative of chitin and the majority of its glucopyranose residues are 2-amino-2-deoxy-β-D-glucopyranose.
2.3.2 Chitosan
Chitin is a semicrystalline polymer with extensive inter- and intra-molecular hydrogen bonding. Hence, it is difficult to dissolve chitin in organic solvents or dilute acids under mild conditions. When chitin is deacetylated by over about 60%, it can easily be dissolved in dilute aqueous acids and is referred to as chitosan or deacetylated chitin. Thus, chitosan is the collective name given to the polymers that are deacetylated from chitin. The only difference between chitosan and chitin is the degree to which the former is deacetylated. Chitosan was first discovered in 1859 by Rouget while boiling chitin with concentrated potassium hydroxide solution that resulted in the deacetylation of chitin. In 1934, Rigby obtained two patents, one for chitosan production from chitin and other for making films and fibres from chitosan. Clark and Smith reported the first X-ray pattern of chitosan fibre. The first review on chitin and chitosan was published by Muzzarelli in 1977. Due to its versatile physical and chemical properties, unique cationic nature, biodegradability, biocompatibility, non-toxicity and antimicrobial activity, chitosan has been extensively investigated for wide range of applications for the last three decades. Chitosan is seen as a new functional material with high application potential in various fields. Chitosan has been found to have applications in areas like food processing, cosmetics, biotechnology, agriculture, fibre formation, pharmaceuticals, medical applications, wastewater treatment, paper production, textile dyeing, printing, finishing and textile ink jet printing.
2.3.3 Production of Chitosan:
Chitosan is a high molecular weight and a linear polymerthat is composed of ß-1,4 linked glucosamine (GlcN) with various quantities of N-acetylated GlcN residues, it is normally obtained by the alkaline deacetylation of chitin extracted from on a boundant source of shell fish exoskeletons. The purpose of deacetylation is to produce chitosan that is readily soluble in dilute acetic acid. It can be seen that chitin is mostly comprised of acetamide groups while chitosan is a copolymer containing acetamide and primary amino groups.

2.3.4 Simplified chitosan production scheme:
Crustacean Shells
↓
Washing and Drying
↓
Demineralisation ←1N HCl for 30 minutes at room temperature with Solid: solvent (1:15, w/v)
↓
Grinding and Sieving
↓
Filtering, Washing and Drying
↓
Deacetylation ← 50% NaOH for 30 minutes at 1000C with solid: solvent (1:10, w/v) in air
↓
Washing and Drying
↓
Chitosan
2.3.5 Physicochemical Characteristics of Chitosan
I. Degree of Deacetylation (DD)
Depending on the production method and species used, the degree of deacetylation ranges from 56% to 99%. For good solubility, the degree of deacetylation should be at least 85%. The degree of deacetylation can be obtained directly by determining amino group content of a chitosan sample or indirectly by determining acetyl content (degree of Nacetylation).
II. Molecular Weight (MW)
The MW of chitin and chitosan varies with the sources and the methods of preparation. Application of chitosan as a textile finish has shown to increase the stiffness of the fabrics, thereby affecting the feel and handle. The effect of MW on the film forming ability of chitosan and on the handle of textile substrates especially cotton will be assessed. The MW of chitosan (160.9)n.
III. Viscosity
Viscosity of chitosan solution is another property that determines its commercial applications and is affected by the degree of deacetylation, molecular weight, concentration, ionic strength, pH, and temperature. The viscosity of chitosan increases with an increase in molecular weight and concentration of chitosan, while it increases with decrease in pH in acetic acid and decreases with decreasing pH in HCl.
IV. Solubility
Water-soluble chitin can be prepared by either homogeneous deacetylation of chitin or homogeneous Nacetylation of chitosan. Water solubility is obtained by homogeneous reaction instead of heterogeneous reaction and only when the DD of chitin is about 0.5. The water-solubility was attributed to the enhanced hydrophilicity due to random distribution of acetyl groups and the destruction of the tightcrystalline structure of chitin. The solubility of chitosan is very important for its commercial applications as a textile finish, fibre or film former and for its chemical modification. Chitosan dissolves in dilute organic and mineral acids by protonation of free amino groups below pH 6.5. Acetic acid is the most preferred solvent for research and applications of chitosan. Generally, the solubility of chitosan and chitin decreases with increasing MW.
V. Colour
The colour of chitin and chitosan is associated with the carotenoid pigment whose main component is astaxanthin. The carotenoids are strongly bound with proteins in the epithelial layer of the exoskeleton of chitin. The carotenoid level in crustacean is very low and varies depending on dietary pigment availability, crustacean size, its maturation, and genetic difference.
2.3.6 Chemical Properties of Chitosan
The chemical properties of chitosan are as follows:
- Linear polyamine.
- Reactive amino groups.
- Reactive hydroxyl groups available.
- Chelates many transitional metal ions.
2.3.7 Biological Properties of Chitosan
The biological properties of chitosan are as follows:
a) Biocompatible
- Natural polymer
- Biodegradable to normal body constituents.
- Safe and non-toxic (the research chitinase is noteworthy in this respect).
b) Binds to mammalian and microbial cells aggressively.
3. MATERIALS AND METHOD
3.1. Materials
To perform this experiment successfully some materials are required such as Fabric, Reactive Dye, Chitosan and Chemicals. Details descriptions of these materials are given below:
a) Fabric: The raw fabric is collected from local market whose Specification are given below.
Specification of Fabric = [(20*26)/(56*60)]*54
b) Dye: The dye which we used of thesis purpose the specification of this dye are given below.
Table: 3.1 Specification of Dye
| Common Name | Reactive Dye |
| Trade Name | NOVACRON R RED TS-3B |
| Company | Huntsman |
| Country | Switzerland |
| Colour | Red |
| Odour | None |
c) Chitosan: The Specification of Chitosan are given below:
Table: 3.2 Specification of Chitosan
| Common name | Chitosan |
| Chemical Name | Poly-(1-4)-2-Amino-2-deoxy-ß-D-Glucan |
| Company name | ZHENGZHOU SIGMA CHEMICAL CO.LTD |
| Origin | CHINA |
| Molecular Formula | (C6H11NO4) n |
| Molecular Weight | (160.9) n |
d) Chemicals: The Specification of all chemicals which was collected for experiment are given below:
Table: 3.3 Specification of Salt
| Common name | Glauber’s salt |
| Chemical Name | Sodium sulfate decahydrate |
| Molecular Formula | Na2SO4·10H2O |
| Appearance | White or colorless monoclinic crystals |
| Solubility | Water soluble |
| Taste | Walty, bitter taste |
Table: 3.4 Specification of Soda Ash
| Common name | Sodium carbonate (also known as washing soda or soda ash) |
| Chemical Name | Sodium salt of carbonic acid |
| Molecular Formula | Na2CO3 |
| Appearance | White powder |
| Characteristics | Water softener, fixation of dye |
| Solubility | Water soluble |
| Procedure | Salt (sodium chloride) and limestone |
Table: 3.5 Specification of Wetting Agent
| Common Name | Wetting Agents |
| Composition | Mixture of surface-active compounds. |
| Appearance | Clear, colorless, slightly viscous liquid |
| PH | 5.5 |
| Specific Gravity at 200C | About 1 g/cm3 |
| Suitable dilute | Very stable in hard water and to salts, alkalis, acids and bleaching agents. |
Table: 3.6 Specification of Leveling Agent
| Common Name | Leveling Agents |
| Chemical basis | Mixture of an addition complexing agent and polymers. |
| Appearance | Coffee Color, low-viscosity liquid |
| PH | About 7.0 |
| Specific gravity at 20οC | About 1.1 |
| Suitable dilute | Cold or warm water |
| General stability | Stable in hard water, Alkalis and acids. |
3.2 Required Machineries
Table: 3.7 Machineries Used.
| Name of Machine | Model | Brand | Origin |
| Infra-Red Lab Dyeing Machine | Supermat | Co-Power | Taiwan |
| Washing and Dry-Cleaning Color Fastness Tester | 415/8 | James H. Heal | UK |
| Crock Master Color Fastness to Rubbing Tester | 670 Hand Driven crock Master | James H. Heal | UK |
| Spectrophotometer | 650 | USA | |
| Color matching cabinet |
3.3 Methods
3.3.1 Padding procedure
Before dyeing the cotton sample fabric is treated with 0.5 %, 1.0%, 1.5%, 2.5% &3.0% chitosans in the padding machine. After padding the sample fabric is dried at 60oC, 30min and then curing is done at 130oC for 5 minutes.

3.3.2 Stock Solution Preparation
During the lab dip preparation a little amount of dyes and others chemicals are required. The measurement of a little amount dyes and chemicals are very complex.1 gm of dye has taken and soluble in 100 ml of warm water and make 1% dye stock solution. Similarly, 5% of Na2CO3 (Sodium carbonate), 10% gluber salt of stock solution have make. By the preparation of stock solution, we can easily take required amount of dyes and chemicals from the stock solution by the following calculation.
Amount of Chemicals = (Total Liquor × Recipe amount) / (1000 × Stock solution%)
Amount of Dyes = (Weight of materials × shade%) / (Stock solution%)
3.3.3 Recipe Formulation
To perform the experiment, there have differences between salt & chitosan evaluation of self-shade (NOVACRON R RED TS FN-3B). For 1% dyes with gluber salt & various amount of chitosan without salt such as 0.5%, 1%,1.5%, 2.50%, &3.0% chitosans are used and analyses with them. The standard recipe is following.
Table: 3.8 Recipe for 1.0% (on the weight of fabric) shade without treatment the fabric with chitosan.
| Ingredients | Without chitosan | Sample-1 | Sample-2 | Sample-3 | Sample-4 | Sample-5 |
| Chitosan | 05% | 1% | 1.5% | 2.5% | 3% | |
| Dyes (%) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Salt (g/l) | 30 | 30 | 30 | 30 | 30 | 30 |
| Soda ash (g/l) | 8 | 8 | 8 | 8 | 8 | 8 |
| Wetting Agent (g/l) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Leveling Agent (g/l) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Sequestering agent | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| M: L | 1:20 | 1:20 | 1:20 | 1:20 | 1:20 | 1:20 |
3.3.4 Dyeing Procedure


At first untreated and chitosan treated sample has collected & take all auxiliaries and dyes from the stock solution in the dye bath and take water to maintain 1:10 liquor ratio therefore fabric is added in dye bath at room temperature and run 10 minutes. Temperatures gradually increase (2o /min) up to 60oC. Now sodium bicarbonate is added in dye bath for fixation of reactive dye from the stock solution of sodium bicarbonate and measure pH of dye bath and run 60 minutes at 60oC. Complete dyeing the dye liquor has drain.
3.3.5 After Treatment
Removal of hydrolysed and unreacted dye from the goods is a vital step after dyeing. The amount of unfixed dye remaining in a cotton fabric dyed with reactive dyes may have to be less than 0.002% owf. Although bleeding out of such a small amount during subsequent washing by the consumer will not significantly alter the depth of shade of the material, it can visibly stain adjacent white goods. This is usually unacceptable. The entire washing operation involves achieving a compromise between the effectiveness of removal of unfixed dye and the cost of the large volumes of water used, including the heating costs. Low liquor ratio washing saves water but gives less dilution of the washing liquors. To remove unfixed dyed required the material is operating at atmospheric pressure. It can possible to carry out soaping at temperatures 80°C for 10 minutes.
3.3.6 Color Fastness to Rubbing Test (ISO 105 X 12)
The fastness test to rubbing used on a variety of fabric to evaluate the transfer of surface dye from the test fabric when it is applied surface friction or rubber against a rough surface. Crock meter & Grey scale are used for this test. To cut the fabric size 10cm × 4cm & The rubbing cloth size 50mm × 50mm. The crocking cloth against which the sample is rubbed is white, bleach, but without finish and undyed cotton fabric. Rubbing time: 10 Cycle/10second.

Working procedure:
- The rubbing arm is attached to the hook.
- Center a piece of abrasive paper, abrasive side up, on the base of the Crock meter.
- The paper’s long dimension should be in the direction of the rubbing.
- The specimen is placed flat and face-up on the abrasive paper. The specimen’s long dimension should be in the direction of the rubbing.
- A piece of test cloth is centered over the finger so that the weave is parallel with the direction of rubbing. Wrap the sides of the cloth evenly around the finger, making sure the cloth is smooth and taut over the finger’s rubbing area.
- The covered finger is then lowered onto the test specimen.
- The cycle counter is reset.
- The handle is turned at the rate of one turn per second for ten times. (Each back and forth motion of the arm equals one stroke and at a speed of 1 stroke/second.)
- After 10 strokes the specimen is removed and compared with the grays. The acceptable range is normally. This visual assessment is done in a color matching cabinet under standard lighting D65 (Artificial Daylight).
3.3.7 Color Fastness to Wash
The resistance of the loss of color of any dyed or printed material to washing is referred to as its wash fastness. Rota Wash, Scissor, Stitch machine are for this test. To cut the Fabric size 10 cm × 4 cm & also Multi fiber fabric. The color fastness to washing is measured by ISO Recommendation No. 1 (ISO 105 C03). A rating of 1(poor) – 5 (excellent) was assigned to each of the six fiber strips on the fabric.
Recipe:
- Soap: 5 g/l
- Soda ash: 2 g/l
- Temperature: (60 ±2)0C
- Time: 30 min
- ML Ratio: 1:50

Working Procedure:
- Stitch the specimen with multi fiber fabric in one side (length direction).
- Weight the specimen and calculate the volume of wash liquor required for ISO method given 150 ml liquor.
- Place the specimen in the container with the wash liquor.
- Run the machine and raise the temperature to 500C and run for 30 min.
- At the end of the test, place the specimen in the beaker and wash with 2 litter distilled water for 2 min & continuously starring the sample. Then washing in running tape water for 2 min. Excess water remove by hand squeezing.
- Dry the composite specimen in incubator, temperature not more than 600C. Then keep in relax for 1 hours.
- Report: Dry the specimen and the change of shade & degree of staining is measured by Grey Scale & Staining Scale.
4. RESULTS AND DISCUSSIONS
4.1. Measurement of Dye Exhaustion
Dye exhaustion may define as the leaving of a dye from the dye bath and attachment to the fiber being dyed. For instance, 70% exhaustion would mean that 70% of the total amount of dye has attached to the fiber, and 30% is still in solution. Dyebath exhaustion can be calculated as the mass of dye taken up by the material divided by the total initial mass of dye in the bath, for a bath of constant volume.
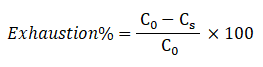
Where, C0 and Cs are the concentrations of dye in the dyebath initially and at some time during the process, respectively.

The treatment of fabric with chitosan has enhanced the dyesite in cellulose macromolecule of cotton fiber. As a result, the treated fabric absorbed more dyestuff than the untreated sample and this absorption has increased the exhaustion percentage of dye in the treated fabric. The improved dyebath exhaustion has shown by the longer bar diagram in Figure 4.1 where all the chitosan treated fabric samples have shown longer bar than the untreated fabric sample. From the statistical analysis, it has been found that, the values of exhaustion percentages of chitosan treated fabric has fluctuated more than the un- treated fabric sample, resulting higher bar in the bar diagram. One possible reason for comparatively higher bar in treated fabric may be the unequal chitosan absorption by different samples.
4.2 Depth of Shade
Color depth of the dyed fabrics was analyzed by measuring the K/S values of samples. Higher the value of K/S more dye will absorb in the fabric. Color measuring instrument (spectrophotometer) determines the K/S value of a given fabric through Kubelka-Munk equation as follows.
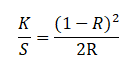
Where, R = reflectance percentage, K = absorption and S = Scattering of dyes. The values found for all the dyed fabrics are shown in Figures 4.2.

It has been found that the chitosan treated fabric samples have absorbed significantly higher amount of dyes than normal untreated fabric. The dyebath parameters such as shade %, amount of salt and alkali, dyeing time and temperature were constant for all the samples. The increased K/S value in the chitosan treated fabric indicates the presence of higher amount of the dye absorbed in the chitosan treated fabric.
4.3 Color Fastness properties
Table: 4.1 Fastness Rating of Rubbing.
| Fabric Type | Rubbing Fastness | |
| Dry | Wet | |
| U | 4/5 | 4 |
| A | 4/5 | 3/4 |
| B | 4/5 | 3/4 |
| C | 4/5 | 3/4 |
| D | 4/5 | 3 |
| E | 4/5 | 3 |
Table: 4.1 Fastness Rating of washing.
| Fabric Type | Washing fastness | Color staining | |||||
| Color change | Acetate | Cotton | Nylon | Polyester | Acrylic | Wool | |
| U | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 |
| A | 5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 |
| B | 5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 |
| C | 5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 | 4/5 |
| D | 4/5 | 4/5 | 4 | 4/5 | 4/5 | 4/5 | 4/5 |
| E | 4/5 | 4/5 | 4 | 4/5 | 4/5 | 4 | 4/5 |
Table: 4.2 Code of test samples
| Test fabric type | Code |
| Normal fabric without any chitosan treatment | U |
| Fabric treated with 0.5% Chitosan solution | A |
| Fabric treated with 1% Chitosan solution | B |
| Fabric treated with 1.5% Chitosan solution | C |
| Fabric treated with 2.5% Chitosan solution | D |
| Fabric treated with 3% Chitosan solution | E |
5. CONCLUSION
5.1 Conclusion
Reactive dyeing process has been completed at 600C and the added time remains 60 mins. Fixation and exhaustion have been completed at 600c. The addition of glubar’s salt is necessary as it increase the exhaustion of the dye bath. This study is intended to improve the absorption of reactive dye by cotton fiber after treating with chitosan. The application of chitosan on cotton fiber enhances the dye adsorption and also increases the hydroxyl group for fixation. Accordingly, dye exhaustion and depth of shade improve in the treated fabric compared to the untreated fabric. Though the fastness rating is lower in case of wet rubbing fastness, the overall fastness rating is satisfactory. In all cases, the pre-dyeing method in which the fabrics were treated with chitosan solutions prior to dyeing produced the optimum K/S value. An increase in chitosan concentration resulted in an increase in the dyeability of dye. This effect was associated with the presence of bonded chitosan through the chemical reaction of the chitosan aldehyde group with cellulose. Due to high absorbency, hydrolysis of dye is reduced which is helpful to protect the environment from pollution. So, the treatment of cotton with chitosan can be an effective way for this purpose.
6. REFERENCES
- Lewin, “Handbook of Fiber Chemistry,” 3rd Edition, Taylor & Francis, New York, 2010.
- Q. Yu, M. Dang and C. W. Yu, “A Preliminary Study on Chemical Degumming of Jute and Kenaf Fibers,” Plant Fiber and Products, Vol. 25, No. 1, 2003, pp. 190- 192.
- Ghosh, A. K. Samanta and G. Basu, “Effect of Selec- tive Chemical Treatments of Jute Fiber on Textile-Re- lated Properties and Processible,” Indian Journal of Fi- bers &Textile Research, Vol. 29, No. 1, 2004, pp. 85-89.
- W.-M. Wang, Z.-S. Cai and J.-Y. Yu, “Study on the Che- mical Modification Process of Jute Fiber,” Journal of En- gineered Fibers and Fabrics, Vol. 3, No. 2, 2008, pp. 1- 11.
- P. G. Gohl and L. D. Vilensky, “Textile Science,” 2nd Edition, Longman Cheshire, New Delhi, 1983.
- A. Shenai, “Chemistry of Dyes and Principles of Dye- ing,” 4th Edition, Sevak, Mumbai, Vol. 2, 1983.
- Broadbent, A. D. (2001). Basic Principles of Textile Coloration. west Yorkshire, UK, England: Sosiety of dyers and Colourists.
- Hustsman. (2004, 06 02). Hustsman Textile Effects. Safety Data Sheet.
- Ingamells, W. (1993). Colour for Textile. West Yorkshire, School of Home Economics and Institutional Management, University of Wales, England: Society of Dyers and Colourists.
- Park, J., & Shore, J. (2004). Practical Dyeing (Vol. 2). West Yorkshire, UK, England: Society of Dyers and Colourists.
- Rebecca, R. B. (2005). Fibre Reactive Dyes with Improved Affinity and Fixation Efficiency.
- Tomasino, D. C. (1992). Chemistry & Technology of Fabric Preparation and Finishing. Raleigh, North Carolina State University, North Carolina: Department of Textile Engineering, Chemistry & Science College of Textile.
- Trotman, E. (1975). Dyeing and Chemical Technology of Textile Fibre (Fifth ed.). High Wycombe, London, England: Charles Griffin & Company Ltd.
- Chitosan and Improved Pigment Ink Jet Printing On Textiles
- Nasar Harun Momin, B. Text. (Textile Chemistry), DKTE’s Textile Engineering Institute, M. Sc. Tech. (Textile Chemistry), University Institute of Chemical Technology, 2003
You may also like:
- Dyeing of Cotton / Polyester Blended Knit Fabric with Reactive / Disperse Dyes Using Jet Dyeing
- Polyester/Cotton (P/C) Blends Fabric Dyeing: Recipe, Parameters, Flowchart and Dyeing Curve
- Blending of Cotton-Polyester Fibre to Produce PC / CVC Yarn
- To Improve the Wettability and Dyeability of Polyester and Polyester Blended (P/C) Fabric by Using Sericin
Founder & Editor of Textile Learner. He is a Textile Consultant, Blogger & Entrepreneur. Mr. Kiron is working as a textile consultant in several local and international companies. He is also a contributor of Wikipedia.






