Fiber Based Biocomposites: Structure, Properties and Applications
Ashish Kumar Dua
M.Tech, Dept. of Textile Engineering,
Indian Institute of Technology (IIT), Delhi.
Email: ashisdua@gmail.com
Biocomposites:
Biocomposites or “Green” composites are fully degradable composites that are made up of biopolymer matrix and natural fibers as reinforcing phase. Biocomposite materials are produced by combining polymer matrix and natural fibers that have their own distinguish properties. However, after combining them, the resultant material possesses superior properties in comparison to those of individual polymer matrix and natural fibers, and they are suitable for various technical applications. Polymer matrix provides the structure and shape of the material, whereas natural fibers improve the performance (tensile, flexural, impact etc.) of the resultant biocomposites. Biocomposite is a new emerging field. Range of polymers has been studied as matrices reinforced with natural fibers. Polymers are synthesized from fossil fuels, bio-based resources and a combination of both. Synthetic polymers include PP, polyethylene, polyvinyls, phenolics and polystyrene.
Need of biocomposites:
- To reduce depletion of petroleum reserves
- To reduce environmental pollution
- To utilize the agricultural waste
- To help attain sustainable development
Reinforcement:
- Reinforcing fibers are cheap and degradable
- Use of agricultural waste like bagasse, oil palm fiber, coir and rice husk etc.
- High amount of fibers/fillers reduces the quantity of matrix used.
- Fibers have disadvantages like aggregation and moisture absorption
- Composites thus made have poor interface and poor impact strength
- The cellulosic fibers have a cell arrangement, divert the crack path by blunting it.
Moisture:
- It hinders the dimensional stability as both matrix and fibers are hydrophillic
- Affinity toward water leads to delamination as the coupling agent no longer bonded to the fiber
- Voids

Fibers used in biocomposites:
- Jute
- Flax
- Ramie
- Hemp
- Kenaf
- Sisal
- Coir
- Oil palm fiber
- Bamboofiber
- Pineapple fiber
- Abaca/banana fiber
- Wood flour
- Sugarcane bagasse and rice husk
Among the lignocellulosic fibers from plant origin, priority of selection is based on availability.

Example,
- Brazil – sugarcane bagasse
- Malaysia, Africa- Oil palm empty bunch fiber
- Mid Asia- hemp, sisal
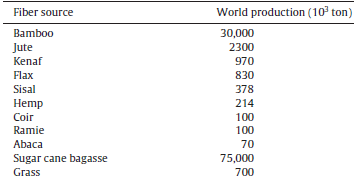
Availability:

Unique about fibers:
Jute-
- Most widely studied lignocellulosic fiber
- Cheapest lignocellulosic fiber in our country
- Has high value of strength 400-800 MPa and modulus 30 Gpa
- Grown for fibers, it is not a waste
- Is not single cell fiber like cotton, so fibrillation can be used after removing bider lignin
Bamboo fiber-
- One of the fastest growing crop, mature in 8 months
- Has spiral angle of 2-10 degree so very strong mechanical properties.
- Has inherent antimicrobial characteristics and easily available
- It is brittle so problem related to uniform processing
- It is reported to absorb UV radiation
Oil Palm Fiber-
- It is one of the large agricultural waste
- For 1 ton of oil, 10 ton fiber waste is generated
- Large quantity is used as fuel or left open in environment
- Has inherent pores on its surface that helps in mechanical interlocking with matrix
- Drawback is the residual oil in the fiber
- There is large variation in diameter
Kenaf-
- Has advantages of very high speed growth. It can grow upto 10cm per day. Mature in 3 months. Moreover it is cultivable in wide variety of weather.
- It suffers from severe agglomeration tendency due to hydrogen bond formation.
Sisal-
- Grown in Brazil and East Asia only
- It is a hard leaf fiber
- Microfibril angle of 10-22° have a tensile strength of 530 MPa
Abaca/Banana-
- Fiber is having a large lumen and are strong fibers.
- These are found to have good saline water resistivity.
- These are strongest among commercially available cellulosic fibers and its composites have better impact properties than flax and jute.
- It is a kind of waste
- Rice husk, coir and bagasse are the agricultural waste and are thus abundant.
Chemical treatment:
Natural fibers allow moisture absorption from the environment which leads to poor bonding with the matrix materials. Chemical treatment modifies surface as well as chemistry of fiber in many cases.
Chemical treat performs 2 functions:
- Removal of binding lignin to open up fibril bundle
- To make the fiber non polar by substituting its hydroxyl group. Thus the fiber is compatible with various hydrophobic resins
Various treatments are as follows…….
Mechanism for natural fibers:
- Most natural fibers are lignocellolosic. Cellulose+lignin+hemicellulose (Hemicell.- D.P =200 Lignin- complex, crosslinked Polymers, binding material)
- Lignin binds the fibrils
- Removing lignin
- Exposes fibrils… Fibrillation
- Surface becomes rough
- Treatment should be peripherial only
Examples,

Alkylation or NaOH treatment:
This involves treatment with NaOH at low conc. 1% to 18% with variable time (2-8 hours)
Fiber – OH + NaOH → Fiber – O – Na + H2O
- Fibers treated at high NaOH concentrations have reduced thermal resistance.
- X-ray studies reveal that order of the crystallite packing increase rather than an increase in the intrinsic crystallinity.
- Surface –OH bonds are modified as there is no peak at 1740 /cm shown in FTIR
- It reduces fiber diameter and uniforms it so that bonding and stress transfer improves.
- Hemp sisal and jute treated at 8% NaOH upto~8hrs shows similar effect where lignin reduces and fiber bundle opens up.
- Only Kapok required high amount 400% for same result.
- Reason is very highly packed crystalline structure of Kapok.
Some results are as follows….

- At low NaOH conc the crystallinity improves but lowers at high conc.
- Cellulose structure changes (I to II)
- Experiments also done on 50% NaOH on coir and 5% NaOH was used for removing residual oil from oil palm fibers.
- Usually at conc 5% or less, time varies from 2 to 72 hours. At elevated temperature fibrillation takes place
Acetylation:
Substitution of surface –OH group by stearic acid group or acetic group.
Process- acetic acid and acetic anhydride individually does not react sufficiently with the fibers → So fibers are initially soaked in acetic acid → treated with acetic anhydride for 1-3 hours at elevated temp.
In another method glacial acetic acid and acetic anhydride treatment is done subsequently with few drops of H2SO4.
Fiber – OH + CH3 – C(=O) – O – C(=O) – CH3 → Fiber – OCOCH3 + CH3COOH
Some results are as follows…….

- Acetylation will make the fibers non-polar and it becomes non reactive to polar resins like cashew nut shell liquid (CNSL).
- When tested on Flax fiber, the strength did not dropped significantly.
- Acetylation is milder treatment than NaOH treatment.
- The porosity of fiber is maintained during acetylation and diameter remains unaffected. Where as reverse in NaOH treatment.
Benzylation:

Other chemical techniques:
- Peroxide treatment
- Sodium chlorite treatment
- Isocyanate treatment
- Permanganate treatment
- Triazine treatment
- Oleoyl chloride treatment
- Fungal treatment
Silane treatment (Coupling agent):
- Silane treatment is done to improve the hydrophillic nature of fiber. Stages of hydrolysis, condensation and bond formation during the treatment.


- Silane coupling agents act as a surface coating.
- This penetrates into the pores and develops mechanically interlocked coatings on fiber surface.
- Silane treated fiber composites provide better tensile strength properties than the alkali treated fiber composites
- Chemical link between the surface of the cellulose fiber and the resin through a siloxane bridge.
Modify fiber surface:
By physical methods – internal chemical structure is intact, only surface topography is changed
- Plasma treatment– role of gas used, pressure of air.
- Corona discharge- Surface itching for short time
- Stretching, calendring, thermo treatment
Bagasse:
Sugarcane bagasse is its residue after extraction. Presently used as
- Fuel
- Paper industry

Composition:
- Cellulose, 56%
- Hemicellulose 6%
- Lignin 29% (possibility of using as matrix?)
- Ash content 7%
- Organic extractive, 2%
Separated fibers are needle like, resembles glass fiber in appearance.

Reason of less research:
Compared with other fibers, sugarcane bagasse fiber has a
- Lower tensile strength
- Lower Young’s’ modulus,
- Higher moisture takeup
- But it has fast biodegradability
Higher contents of hemicellulose (27%) and lignin (21%), (jute fiber- 13 % and 13%, Sisal fiber -13% and 11%)
Fiber separation:
Adv- fibers are readily powedered and fibrillated
Hammer milled → screened using mesh → oven dried → repeated water and alkali wash.
Cut in pieces → washed in water at 70oC → removal of soluble compounds → transformed into pulp in blender → washed with distilled water in sieve mesh → again washed at 70oC to removal small particles, medulla etc → dried until constant weight → organic solvents removed by soxhlet extraction → 48 hrs using cycle hexane and ethanol (1:1 by volume)

Alkali treatment:

- The lignin content of sugar cane bagasse is high, 21% on the average
- It increases the resistance to chemical and microbial attack
- Reported to have very good compatibility with PLA without treatment
- Caro’s acid (persulfuric acid) treatment to remove lignin. 2 and 3 step process
- Benzoic acid was an effective adhesion promoter for BF/PVC composites
Acetylation on bagasse:
There are various ways reported.
- At room temperature using a mixture of glacial acetic acid, toluene, perchloric acid and anhydride acetic, for 48hr.
- Mixture of acetic acid, anhydride acetic for 2hr up to 48hr.
- Higher temperatures were adopted using glacial acetic acid, sulfuric acid and anhydride acetic, at 35oC, followed by anhydride acetic addition at 18oC and 55oC for 5hr.
Experimental:
- 1% NaOH treatment at 25 c for 1 hour is better than other conc.
- Readily attaches with PLA and polyhydroxylbutyrate (PHB), without modification.
- Flexural strength of PHB increased by 50% ,reinforcing the polymer with 30% v/v washed and acetone-treated bagasse fibers
- Poly[methylene (polyphenyl isocyanate)] (PMPPIC), succinic anhydride are successfuly used as coupling agents.
- Optimum mass fraction is 40% to 60% for good impact resiatance.
- Very few literature with biodegradable matrices
Oil Palm Fiber:

- It is one of the large agricultural waste
- For 1 ton of oil,10 ton fiber waste is generated
- Large quantity is used in making pottasium fertilizer or as fuel or left open in environment
- Has inherent pores on its surface that helps in mechanical interlocking with matrix
- There is large variation in diameter
- Drawback is the residual oil in the fiber.


- India is a major Importer despite large production Agricultural ministry-North east and southern India.
Morphology and properties:
- It is hard and tough similar to coir fiber
- Porous surface morphology (also facilitates penetration of water)

- The major constituents of these fibers were found to be cellulose. Lignin content is comparatively less
- Controversy over the fibrillar structure
- Starch and silica granules present. Silica can be removed mechanically to give clear pores.
- Cellulose 42.7–65%, lignin 13-20%, hemicell-17-33%
- Strength is comparable to coir
- The pores became more prominent upon alkali treatment 5% NaOH for 48 hours, 25oC temperature
Experimental:
| Treatment | Effect |
| Mercerization | Amorphous waxy cuticle layer leaches out |
| Gamma irradiation | Partially eliminates the porous structure of the fiber and causes microlevel disintegration. It degrades mechanical properties. |
| Silane treatment | Imparts a coating on fiber surface |
| Acetylation | Removes waxy layer from the surface and makes the fiber hydrophobic |
| Alkali treatment | Makes the surface pores wider and fiber become thinner due to dissolution of natural and artificial impurities |
| Oil extraction | Bright colour to the fiber. Removal of oil layer exposes surface pits and makes surface coarse |
Silane treatment (1% silane solution (tri- ethoxy vinylsilane) in water–ethanol mixture (40 : 60) for 3 h. pH 3.5–4) gives better result than alkali treatment
Various coupling agents used for it
- Epolene (maleic anhydride modified-polypropylene)
- Poly[methylene (polyphenyl isocyanate)] (PMPPIC)
- γ –methacryloxypropyltrimethoxysilane
- Vinyltri (2-methoxy ethoxy) silane
- γ –aminopropyltriethoxysilane
- Poly(propylene-acrylic acid)
- Poly (propylene-ethylene acrylic acid)
Areas yet to explore (from reference paper):
- Effect of OPF size on the composite properties
- Information on thermal properties, electrical resistance, high voltage break down characteristics,
- Degradation/weathering characteristics, deflection in thermal environment, rheology, resistance to various chemicals etc.
- OPF compatibility and mechanical properties with biodegradable matrices.
Matrices:
- Matrices from natural resources are mostly hydrophillic.
- Do not require Coupling agent to compatiblize the interface
- starch, Soy protein, PLA , lignin from wood waste etc.
- Disadvantage is their Brittle nature and low processing temperature
PLA-polylactic acid:
- Has high potential as matrix as it can be manufactured in large scale once the prodution technology successfuly invented.
- PLA is brittle (and its blend with startch is still brittle)
- PLLA m.p 170-183oC, Tg- 55-65oC density 1.25 g/cc PDLLA Tg 59oC. PLLA is crystalline in nature, PDLLA is fully amprphous..
- Kenaf and PLA composites are widely studied.
- Decomoses by hydrolitic cleavage by ester group then chain breaks to smaller chain and monomers. Most chain ends are carboxylic that autocatalyse the hydrolysis step
SOY:
Abundant and used for oil and protein suppliment. As rich in protein, it has various functional groups besides amino and carboxyllic groups. It is also brittle in nature. Its major components, 11s and 7 s are reported to be capable of polymerisation
- Soy flour- 50% protein
- SPC or soy protein composites – 60% protein + carbohydrates
- SPI or soy protein integrate – 90% protein and is refined form of SPC
- Soy milk
You may also like:
- What is Composite | Textile Structural Reinforced Composites
- Analyzing the Effect of Mechanical Properties on Chicken Feather Reinforced Polypropylene Composites
- Acoustic Absorption Performance of Natural Fibers & Their Reinforced Composites
- Green Composites: Types, Properties and Manufacturing Process
- Glass Fiber Composites: Properties, Manufacturing and Applications
- Different Important Functions of Geotextile
- Application of Textile Reinforced Concrete in Sustainable Construction
Founder & Editor of Textile Learner. He is a Textile Consultant, Blogger & Entrepreneur. Mr. Kiron is working as a textile consultant in several local and international companies. He is also a contributor of Wikipedia.






